- Reaction score
- 950
Happy new year, baldites! I just discovered this recent patent out of Stanford University for creating artificial peptides that act as powerful Wnt agonists.

The peptides consist of at least three components. The first binds to the Wnt receptor Frizzled and the second binds to the Wnt co-receptor Lrp5/6 - these two parts mimic the action of Wnt proteins. The third component is an R-spondin agonist to amplify the Wnt signal - this is important as I'll explain later.
The patent discusses using this to treat, among many other conditions, androgenetic alopecia.

So why is Wnt signaling important for those of us with A.G.A?
Well, for one thing, there's strong genetic evidence for a causal link between Wnt signaling and A.G.A. This is from the most recent GWAS (genome-wide association study) on A.G.A:
The basic scheme for a GWAS (genome-wide association study) is the following:
1. Take a group of people with some condition (in this case balding men), another group without that condition (in this case fullheads).
2. Look for statistically significant genetic differences between them.
3. Genetic differences between the groups will usually be found near genes that contribute to that condition.
In the case of A.G.A, many genetic differences between bald men and fullheads are found near Wnt-related genes. Here's a representation of some (not all) Wnt-related genes associated with A.G.A and their roles in the pathway, using results from four different GWAS (Hagenaars et al., 2017) (Heilmann-Heimbach et al., 2017) (Pickrell et al., 2016) (Pirastu et al., 2017):
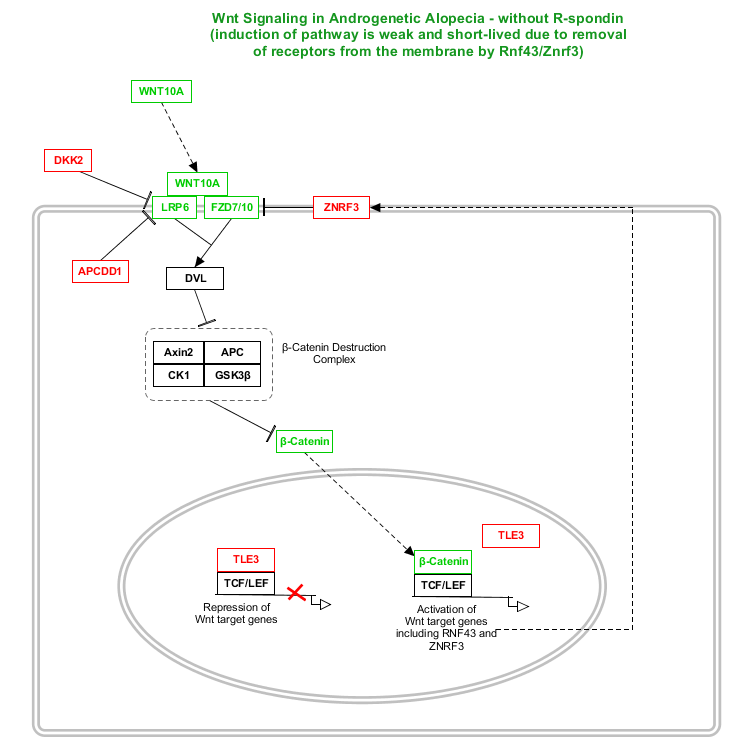
A.G.A-associated genes that are positive regulators of Wnt signaling are green, while A.G.A-associated genes that are negative regulators of Wnt signaling are red. Other Wnt-related genes are black. A quick explanation of how this pathway works:
1. Beta-catenin has two major roles in the cell - the first is in regulating cell-cell adhesion, and the second is as a transcription factor (a transcription factor is a protein that regulates transcription of genes). We're concerned with the second of these.
2. When Wnt signaling is off, a protein complex consisting of TCF or LEF and so-called "Groucho" proteins bind to DNA and repress transcription of many genes. One Groucho family member, TLE3, is associated with A.G.A. A destruction complex consisting of Axin2, APC, CK1, and GSK3-beta tags beta-catenin (an A.G.A-associated gene) for degradation.
3. To turn Wnt signaling on: A Wnt protein (like WNT10A - associated with A.G.A) secreted from a nearby cell binds to the Wnt receptor called "Frizzled" (two separate Frizzled receptors -- FZD7 and FZD10 -- are associated with A.G.A). Wnt also binds to the Wnt co-receptor LRP5 or LRP6 (LRP6 is associated with A.G.A). Now we have a protein complex consisting of Wnt, Frizzled, and Lrp5/6 - an activated Wnt signaling complex.
4. The activated Wnt signaling complex directs the protein Disheveled (DVL) to disable the beta-catenin destruction complex. Now beta-catenin is free to accumulate and move into the nucleus.
5. A large number of beta-catenin proteins are now in the nucleus, where they bind to TCF/LEF. When beta-catenin binds to TCF/LEF instead of Groucho, the TCF/LEF-beta-catenin complex now activates Wnt target genes rather than repressing them. Activation of Wnt target genes is essential for hair growth. However, many Wnt target genes are also negative feedback regulators of the pathway (like Axin2, a member of the beta-catenin destruction complex). We'll focus particularly on two other negative regulators of the Wnt pathway - RNF43 and the A.G.A-associated gene ZNRF3. RNF43/ZNRF3 tag Frizzled receptors (including the newly-activated complexes) for removal from the cell membrane and subsequent degradation. Soon after its initial activation, the Wnt signal is gone. Under such conditions, Wnt signaling is weak and short-lived. Keep Rnf43/Znrf3 in mind for later.
Hopefully this convinces you of the importance of Wnt signaling in A.G.A. Now, what happens when Wnt signaling is repressed in hair folllicles? Something closely approximating A.G.A, as shown by experiments where A.G.A-associated gene WNT10A is knocked out in mice (Xu et al., 2017).
The results of the study by Xu et al. are consistent with previous observations that:
- Wnt signaling controls dermal papilla size and hair follicle size, hence the miniaturization. (Lei et al., 2014, among others)
- Blocking Wnt signaling in hair follicle stem cells causes them to differentiate into sebocytes (the cells that make up the sebaceous gland) rather than hair follicle cells (Lien et al., 2014, among others)
- Wnt signaling is required for conversion of hair follicle stem cells into Lgr5+ progenitor cells capable of making an entire hair follicle (Hoeck et al., 2017) (Jaks et al., 2008)
Remember: The peptides described in this latest patent have two components to mimic the action of Wnt proteins - one to bind Frizzled and one to bind Lrp5/6. This forms an activated Wnt signaling complex just as Wnt proteins would.
Now - remember Rnf43/Znrf3? Many Wnt-dependent organs, including hair follicles, have a way of blocking the action of these two negative Wnt regulators. Here it is:
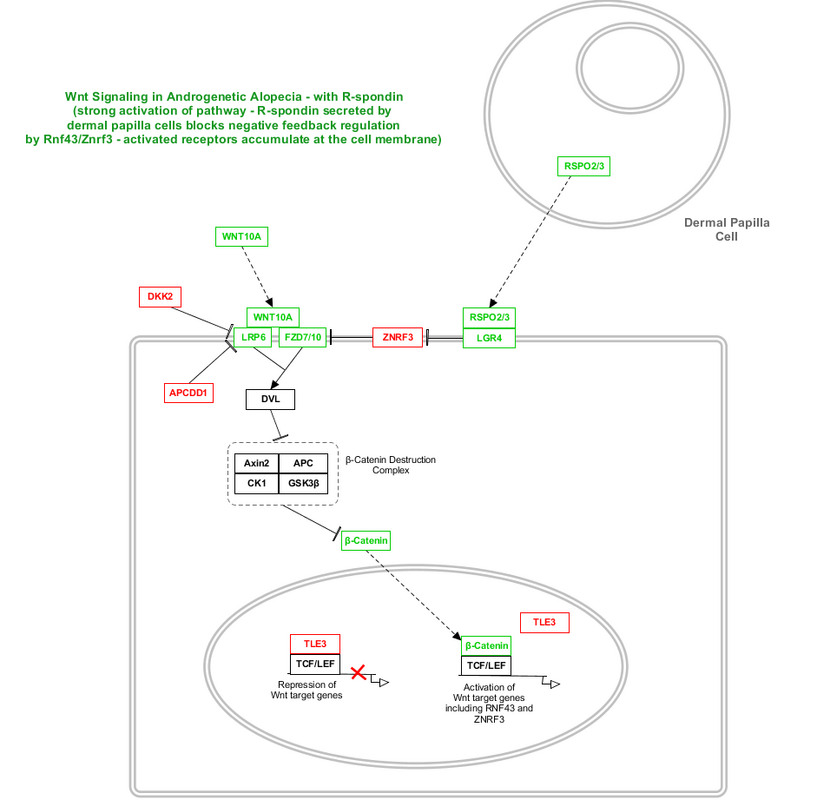
1. Wnt/beta-catenin activates expression of RNF43 and A.G.A-associated ZNRF3. But something different happens in this case.
2. R-spondins (two of which are associated with A.G.A - RSPO2 and RSPO3) -- secreted from dermal papilla cells -- bind to their receptor LGR4/5/6 (LGR4 is associated with A.G.A).
3. The RSPO/LGR4 complex binds ZNRF3 and prevents it from interacting with Frizzled receptors.
4. Now, activated Wnt complexes accumulate at the cell membrane. Wnt signaling is now very strong and long-lasting.
The association of A.G.A with RSPO2 is especially strong, with multiple large-effect variants near the RSPO2 (R-spondin2) gene. This pattern is also found around the Androgen Receptor (AR) gene and to a lesser extent the SRD5A2 (5-alpha reductase type II) gene. In other words, we should expect that R-spondin2 plays a particularly important role in A.G.A.
Anyway, here's an animation and explanation by Hans Clevers (an expert on the Wnt pathway) of this (from 10:00-12:45 only, not the whole thing):
The third component of this peptide is an R-spondin agonist. To give you a taste for just how powerful this is, look at these figures from the patent.
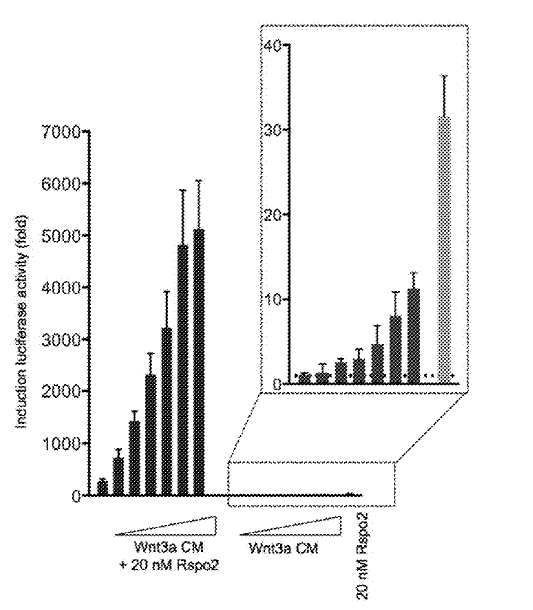
What this shows is activity of an artificial gene called TOPFlash, which gives a readout of Wnt signaling activity. In this first one, the highest concentration of Wnt3a alone induces a 10-fold increase in TOPFlash activity compared to control. Wnt3a plus R-spondin2 induces a 5,000-fold increase in TOPFlash activity (i.e. it enhances Wnt signaling by 500 times compared to Wnt alone).
This next one shows the activity of a peptide containing only Frizzled and Lrp5/6 binding domains (a Wnt-only agonist) compared to peptides additionally containing an R-spondin agonist domain.
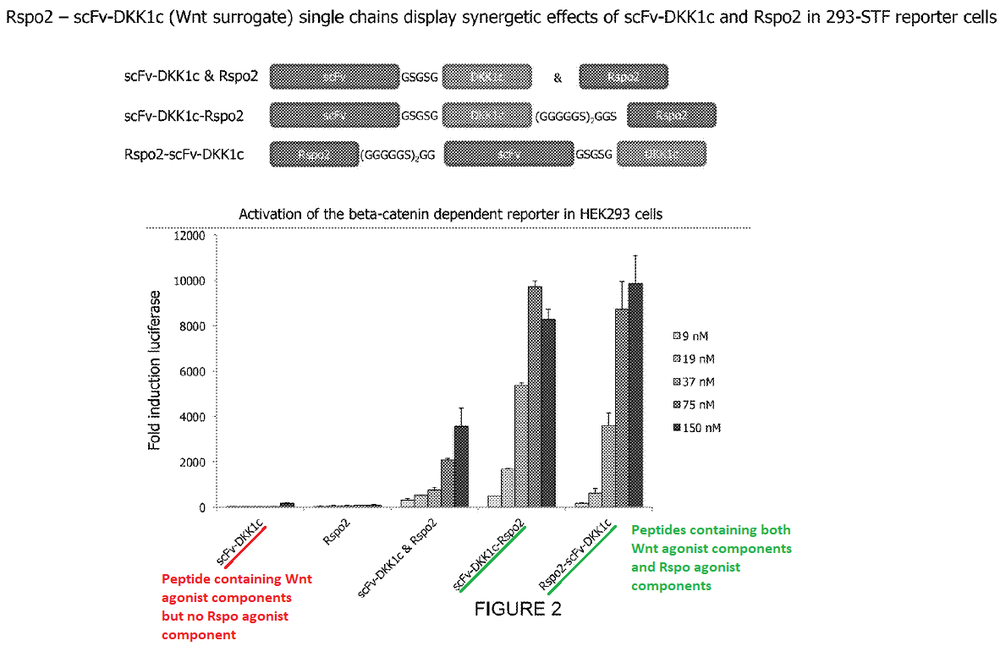
Also in this case, the combined Wnt + R-spondin agonist peptides tower over the Wnt-only agonist peptide.
This massive amplification is not just trivia but, as Hans Clevers mentioned in the video, functionally important. For example, the combination of Wnt + R-spondin can maintain and expand intestinal stem cells, while Wnt alone cannot (Yan et al., 2017). Intestinal stem cells quickly die when the R-spondin receptor LGR4 is knocked out. Wnt completely fails to prevent this. The GSK3-beta inhibitor lithium chloride does slightly better, but still rescues just 6% of the cells (Mustata et al., 2011).
Is robust hair growth also dependent on R-spondins? Some early evidence says Yes. The following, from Smith et al., 2016, shows cultured human hair follicles treated with nothing, Wnt3a alone, and Wnt3a + R-spondin2 respectively.
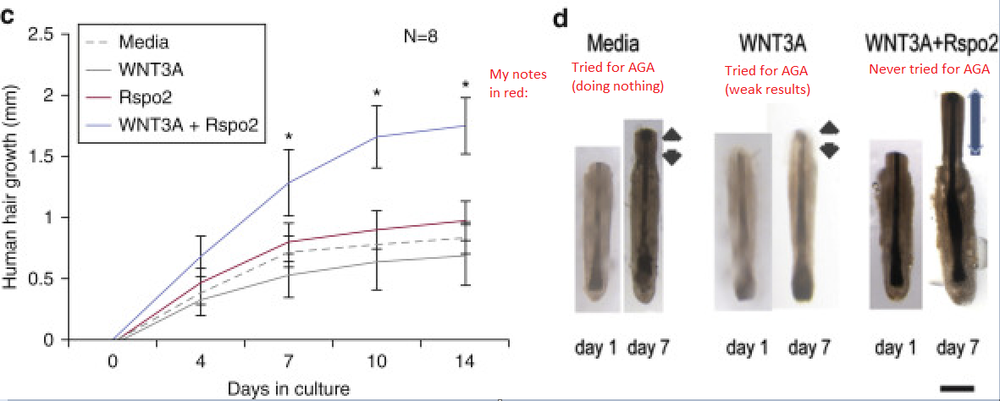
The graph shows that Wnt3a + Rspo2 treated follicles grew much faster than either controls or follicles treated with Wnt3a alone. Also, at least in the example shown, the hair shaft looks like it may have more than doubled in thickness.
In another experiment, cultured hair follicle stem cells were treated with Wnt3a alone, Wnt3a + R-spondin1, R-spondin1 alone, or nothing. While R-spondin1 enhanced expression of several Wnt target genes as expected, expression of Lgr5 was completely dependent on R-spondin.
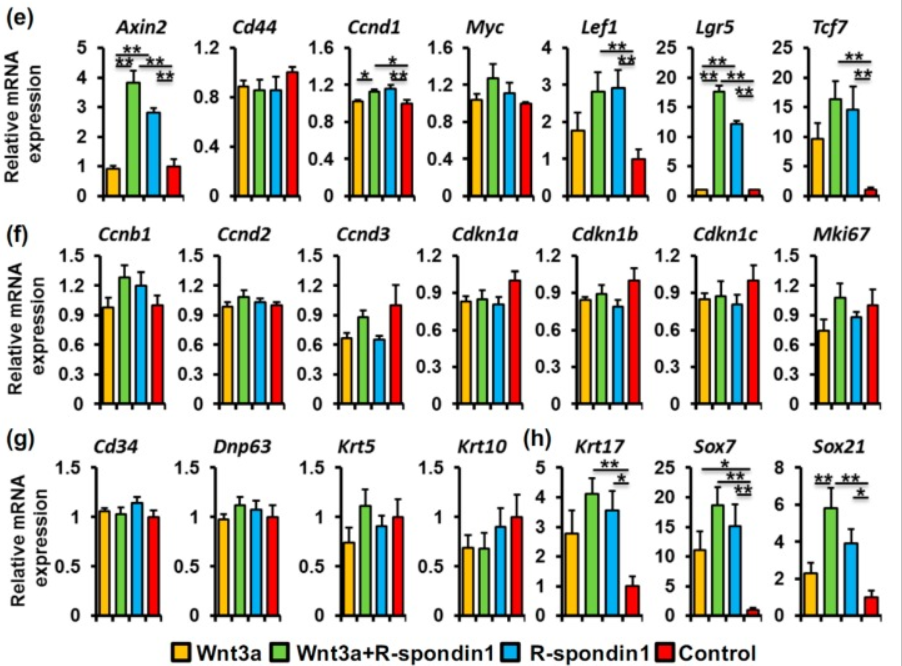
Lgr5 (itself an R-spondin receptor of course) is a marker for a type of hair follicle progenitor cell that is missing in bald scalp (Garza et al., 2011). Conversion of hair follicle stem cells to these Lgr5+ progenitors requires Wnt signaling, and these cells can form entirely new hair follicles, as I mentioned earlier.
Last but not least: At the recent Hair Congress (thanks hellouser), Jeff Biernaskie showed that dermal stem cells express LGR4, and that R-spondin from nearby dermal papilla cells induces those cells to proliferate. Dermal stem cells can make new dermal papilla cells. Therefore, we could speculate that R-spondin might play some role in expanding the dermal stem cell population, and perhaps in activating dermal stem cells to make new dermal papilla cells. It is after all true that Wnt signaling controls dermal papilla size. Since the size of the dermal papilla controls the size of the hair shaft (Chi et al., 2013), and since in A.G.A the dermal papilla has become very small and the hair shaft very thin, enlarging the dermal papilla is key to treating A.G.A.
So IMO, the patented Wnt/R-spondin agonist has solid potential for treating A.G.A. It can potentially:
- extend the anagen phase of the hair cycle (the paper by Smith et al. demonstrates this)
- enlarge the dermal papilla and consequently the hair shaft
- convert hair follicle stem cells to Lgr5+ hair follicle progenitors, and stop them from making sebocytes instead
All of these are critical for treating A.G.A. The patented agonist would also be useful for wound-induced hair follicle neogenesis, since Wnt is the earliest signal in hair follicle induction. Fingers crossed that they actually move forward with this.
The peptides consist of at least three components. The first binds to the Wnt receptor Frizzled and the second binds to the Wnt co-receptor Lrp5/6 - these two parts mimic the action of Wnt proteins. The third component is an R-spondin agonist to amplify the Wnt signal - this is important as I'll explain later.
The patent discusses using this to treat, among many other conditions, androgenetic alopecia.
So why is Wnt signaling important for those of us with A.G.A?
Well, for one thing, there's strong genetic evidence for a causal link between Wnt signaling and A.G.A. This is from the most recent GWAS (genome-wide association study) on A.G.A:
The selected genes were tested for enrichment of known pathways and gene sets using ConsensusPathDB-human22. Many pathways known to affect MBP were enriched, in particular Wnt signalling and apoptosis (Supplementary Data 12).
Three main groups were found: genes linked to Wnt signalling (LGR4, RSPO2, WNT3, WNT10A, SOX13, DKK2, TWIST1, TWIST2, IQGAP1 and PRKD1), genes involved in apoptosis (BCL2, DFFA, TOP1, IRF4 and MAPT) and a third more heterogeneous group including the androgen receptor and TGF-beta pathways (RUNX2, RUNX3, PTHLH, ALPL, AR, RUNX1, PDGFA, SRD5A2, FGF5 and PAX3). Supplementary Fig. 1 shows the membership of each gene in each pathway. Although many different pathways have been implicated in the development of MBP25, our results suggest that in addition to the androgen receptor pathway, for which we confirm a prominent function, the Wnt and apoptosis pathways play central roles. male pattern baldness is characterised by a shorter growth (anagen) phase, which has been associated with increased apoptosis25of the hair follicle cells. These results suggest that the anagen phase becomes shorter because of differences in the genes regulating apoptosis. The Wnt pathway has been implicated in the transition from the resting (telogen) phase to the anagen phase26, and also in the determination of the fate of the stem cells in the hair bulge27, which are both dysregulated in balding tissue.
The basic scheme for a GWAS (genome-wide association study) is the following:
1. Take a group of people with some condition (in this case balding men), another group without that condition (in this case fullheads).
2. Look for statistically significant genetic differences between them.
3. Genetic differences between the groups will usually be found near genes that contribute to that condition.
In the case of A.G.A, many genetic differences between bald men and fullheads are found near Wnt-related genes. Here's a representation of some (not all) Wnt-related genes associated with A.G.A and their roles in the pathway, using results from four different GWAS (Hagenaars et al., 2017) (Heilmann-Heimbach et al., 2017) (Pickrell et al., 2016) (Pirastu et al., 2017):
A.G.A-associated genes that are positive regulators of Wnt signaling are green, while A.G.A-associated genes that are negative regulators of Wnt signaling are red. Other Wnt-related genes are black. A quick explanation of how this pathway works:
1. Beta-catenin has two major roles in the cell - the first is in regulating cell-cell adhesion, and the second is as a transcription factor (a transcription factor is a protein that regulates transcription of genes). We're concerned with the second of these.
2. When Wnt signaling is off, a protein complex consisting of TCF or LEF and so-called "Groucho" proteins bind to DNA and repress transcription of many genes. One Groucho family member, TLE3, is associated with A.G.A. A destruction complex consisting of Axin2, APC, CK1, and GSK3-beta tags beta-catenin (an A.G.A-associated gene) for degradation.
3. To turn Wnt signaling on: A Wnt protein (like WNT10A - associated with A.G.A) secreted from a nearby cell binds to the Wnt receptor called "Frizzled" (two separate Frizzled receptors -- FZD7 and FZD10 -- are associated with A.G.A). Wnt also binds to the Wnt co-receptor LRP5 or LRP6 (LRP6 is associated with A.G.A). Now we have a protein complex consisting of Wnt, Frizzled, and Lrp5/6 - an activated Wnt signaling complex.
4. The activated Wnt signaling complex directs the protein Disheveled (DVL) to disable the beta-catenin destruction complex. Now beta-catenin is free to accumulate and move into the nucleus.
5. A large number of beta-catenin proteins are now in the nucleus, where they bind to TCF/LEF. When beta-catenin binds to TCF/LEF instead of Groucho, the TCF/LEF-beta-catenin complex now activates Wnt target genes rather than repressing them. Activation of Wnt target genes is essential for hair growth. However, many Wnt target genes are also negative feedback regulators of the pathway (like Axin2, a member of the beta-catenin destruction complex). We'll focus particularly on two other negative regulators of the Wnt pathway - RNF43 and the A.G.A-associated gene ZNRF3. RNF43/ZNRF3 tag Frizzled receptors (including the newly-activated complexes) for removal from the cell membrane and subsequent degradation. Soon after its initial activation, the Wnt signal is gone. Under such conditions, Wnt signaling is weak and short-lived. Keep Rnf43/Znrf3 in mind for later.
Hopefully this convinces you of the importance of Wnt signaling in A.G.A. Now, what happens when Wnt signaling is repressed in hair folllicles? Something closely approximating A.G.A, as shown by experiments where A.G.A-associated gene WNT10A is knocked out in mice (Xu et al., 2017).
By 6 months of age, Wnt10a mutant HFs became miniaturized with enlarged sebaceous glands and elevated lipid production (Fig. 5n–s). Dominant negative Lef1 also causes sebaceous gland expansion31, consistent with decreased Wnt/β-catenin signalling in Wnt10a mutant HFs. Despite HF miniaturization in Wnt10a mutants, CD34+ KRT15+ bulge stem cells were retained (Fig. 5t–w). Miniaturized HFs in human androgenetic alopecia similarly display enlarged sebaceous glands and bulge stem cell retention32. As data from genome-wide association studies indicate association of a WNT10A variant with androgenetic alopecia6, decreased WNT10A/β-catenin signalling may contribute to this condition.
Using genetic mouse models, we show that long-term absence of WNT10A causes HF miniaturization and sebaceous gland enlargement, with retention of bulge stem cells. This phenomenon is also observed in human androgenetic alopecia, consistent with identification of a WNT10A variant associated with lower expression levels and male pattern baldness. Functional studies of the WNT10A variant may shed further light on the aetiology of this common condition.
The results of the study by Xu et al. are consistent with previous observations that:
- Wnt signaling controls dermal papilla size and hair follicle size, hence the miniaturization. (Lei et al., 2014, among others)
- Blocking Wnt signaling in hair follicle stem cells causes them to differentiate into sebocytes (the cells that make up the sebaceous gland) rather than hair follicle cells (Lien et al., 2014, among others)
- Wnt signaling is required for conversion of hair follicle stem cells into Lgr5+ progenitor cells capable of making an entire hair follicle (Hoeck et al., 2017) (Jaks et al., 2008)
Remember: The peptides described in this latest patent have two components to mimic the action of Wnt proteins - one to bind Frizzled and one to bind Lrp5/6. This forms an activated Wnt signaling complex just as Wnt proteins would.
Now - remember Rnf43/Znrf3? Many Wnt-dependent organs, including hair follicles, have a way of blocking the action of these two negative Wnt regulators. Here it is:
1. Wnt/beta-catenin activates expression of RNF43 and A.G.A-associated ZNRF3. But something different happens in this case.
2. R-spondins (two of which are associated with A.G.A - RSPO2 and RSPO3) -- secreted from dermal papilla cells -- bind to their receptor LGR4/5/6 (LGR4 is associated with A.G.A).
3. The RSPO/LGR4 complex binds ZNRF3 and prevents it from interacting with Frizzled receptors.
4. Now, activated Wnt complexes accumulate at the cell membrane. Wnt signaling is now very strong and long-lasting.
The association of A.G.A with RSPO2 is especially strong, with multiple large-effect variants near the RSPO2 (R-spondin2) gene. This pattern is also found around the Androgen Receptor (AR) gene and to a lesser extent the SRD5A2 (5-alpha reductase type II) gene. In other words, we should expect that R-spondin2 plays a particularly important role in A.G.A.
Anyway, here's an animation and explanation by Hans Clevers (an expert on the Wnt pathway) of this (from 10:00-12:45 only, not the whole thing):
The third component of this peptide is an R-spondin agonist. To give you a taste for just how powerful this is, look at these figures from the patent.
What this shows is activity of an artificial gene called TOPFlash, which gives a readout of Wnt signaling activity. In this first one, the highest concentration of Wnt3a alone induces a 10-fold increase in TOPFlash activity compared to control. Wnt3a plus R-spondin2 induces a 5,000-fold increase in TOPFlash activity (i.e. it enhances Wnt signaling by 500 times compared to Wnt alone).
This next one shows the activity of a peptide containing only Frizzled and Lrp5/6 binding domains (a Wnt-only agonist) compared to peptides additionally containing an R-spondin agonist domain.
Also in this case, the combined Wnt + R-spondin agonist peptides tower over the Wnt-only agonist peptide.
This massive amplification is not just trivia but, as Hans Clevers mentioned in the video, functionally important. For example, the combination of Wnt + R-spondin can maintain and expand intestinal stem cells, while Wnt alone cannot (Yan et al., 2017). Intestinal stem cells quickly die when the R-spondin receptor LGR4 is knocked out. Wnt completely fails to prevent this. The GSK3-beta inhibitor lithium chloride does slightly better, but still rescues just 6% of the cells (Mustata et al., 2011).
Is robust hair growth also dependent on R-spondins? Some early evidence says Yes. The following, from Smith et al., 2016, shows cultured human hair follicles treated with nothing, Wnt3a alone, and Wnt3a + R-spondin2 respectively.
The graph shows that Wnt3a + Rspo2 treated follicles grew much faster than either controls or follicles treated with Wnt3a alone. Also, at least in the example shown, the hair shaft looks like it may have more than doubled in thickness.
In another experiment, cultured hair follicle stem cells were treated with Wnt3a alone, Wnt3a + R-spondin1, R-spondin1 alone, or nothing. While R-spondin1 enhanced expression of several Wnt target genes as expected, expression of Lgr5 was completely dependent on R-spondin.
We further studied the effect of R-spondin1 on bulge stem cells in vitro to help find possible mechanism(s) through which exogenous R-spondin1 injection promote anagen entry. It is reported that LGR5 is strongly expressed in the lower portion of the CD34+ bulge stem cells in telogen [15], but the cultured bulge stem cell line we used in the in vitro assays which was originally derived from telogen mouse back skin based on CD34 expression [16] did not show LGR5 expression. It is possible that the LGR5 expression is lost during the long-time amplification in vitro. Nevertheless, these bulge stem cells express another R-spondin1 receptor, LGR4, which, we believe, makes them responsive to R-spondin1. In our in vitro study, R-spondin1 supplementation alone exhibits a considerable effect on the enhancement of Wnt/β-catenin activity, indicating the existence of endogenous Wnt ligands. Indeed, we detected there were the expression of several Wnt ligands, either in bulge stem cells or in the feeder cells, such as Wnt3a, Wnt4, Wnt5, and Wnt10a. Therefore, we pondered R-spondin1 might exaggerate the Wnt/β-catenin activity in the presence of some of these Wnt ligands.
Several of the Wnt target genes that were upregulated by R-spondin1 (or together with Wnt3a) in the bulge stem cells in vitro, such as Axin2 and Lef1, are reported to be associated with HFSC activation in vivo upon telogen-to-anagen transition [35]. LGR5, which marks proliferative, but not quiescent, stem cells in the gastrointestinal tract and the hair follicle is also a Wnt target, per se. Intriguingly, as mentioned above, LGR5 was initially absent in the bulge stem cells in vitro, but it showed a drastic upregulation with R-spondin1 treatment (but not with Wnt3a alone), implying the potential importance of R-spondin1 in HFSC activation. We did not detect evident changes in the expressions of genes associated with cell proliferation (Ccnb1, Ccnd1/2/3, Mki67, etc.), but substantial change in HFSC fate related genes which are also Wnt/β-catenin targets (Krt17, Sox7, and Sox21) occurred. Based on these data, it is speculated that R-spondin1-mediated Wnt/β-catenin activation does not directly regulate HFSC proliferation in vitro, but possibly control HFSC fate determination and in turn initiates HFSC activation.
Lgr5 (itself an R-spondin receptor of course) is a marker for a type of hair follicle progenitor cell that is missing in bald scalp (Garza et al., 2011). Conversion of hair follicle stem cells to these Lgr5+ progenitors requires Wnt signaling, and these cells can form entirely new hair follicles, as I mentioned earlier.
Last but not least: At the recent Hair Congress (thanks hellouser), Jeff Biernaskie showed that dermal stem cells express LGR4, and that R-spondin from nearby dermal papilla cells induces those cells to proliferate. Dermal stem cells can make new dermal papilla cells. Therefore, we could speculate that R-spondin might play some role in expanding the dermal stem cell population, and perhaps in activating dermal stem cells to make new dermal papilla cells. It is after all true that Wnt signaling controls dermal papilla size. Since the size of the dermal papilla controls the size of the hair shaft (Chi et al., 2013), and since in A.G.A the dermal papilla has become very small and the hair shaft very thin, enlarging the dermal papilla is key to treating A.G.A.
So IMO, the patented Wnt/R-spondin agonist has solid potential for treating A.G.A. It can potentially:
- extend the anagen phase of the hair cycle (the paper by Smith et al. demonstrates this)
- enlarge the dermal papilla and consequently the hair shaft
- convert hair follicle stem cells to Lgr5+ hair follicle progenitors, and stop them from making sebocytes instead
All of these are critical for treating A.G.A. The patented agonist would also be useful for wound-induced hair follicle neogenesis, since Wnt is the earliest signal in hair follicle induction. Fingers crossed that they actually move forward with this.
