Let's start with a quick overview of the problem. Bald scalp does not lack hair follicle stem cells, but these HFSCs don't express the necessary markers to differentiate into hair-producing cells.
 www.ncbi.nlm.nih.gov
www.ncbi.nlm.nih.gov
There's a lack of CD34+ and LGR5+ cells in bald scalp. After injury, an inflammatory response, involving NF-KB and Myc, causes CD34+ cells to divide. In the presence of Wnt signaling these and other stem cells are converted to LGR5+ cells. CD34+ cells are not required for hair growth. Their deletion has been shown not to prevent hair growth, but LGR5 is required for hair growth.
It's evident that Wnt target genes are required for hair growth and are downregulated in bald scalp.
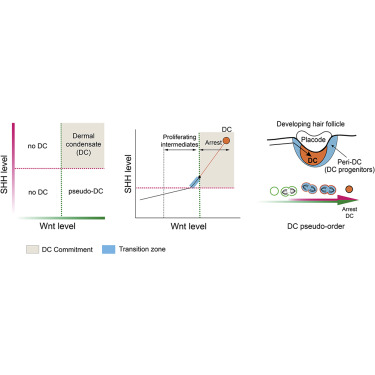
In fact, a new study finds that the only signals required for hair growth are Shh and Wnt.
 www.sciencedirect.com
www.sciencedirect.com
 yaledailynews.com
yaledailynews.com
Unfortunately both of these pathways are involved in tumorigenesis. The highly potent Smoothened(Shh) agonists, SAG and purmorphamine are too risky to use. There is a group of glucocorticoids that have been shown to potently induce Shh, and have been used regularly without causing cancer. Halcinonide is the most potent. That leaves Wnt activation. What compounds can be used for that? Direct Wnt receptor agonists are out of the question. Exogenous Wnt proteins have been shown to cause cancer in mice, just like SAG. In limited testing R-spondin did not have this effect, and it upregulates Wnt signaling only where it is supposed to be upregulated.
Uniformly upregulating Wnt signaling using GSK3b inhibitors or SFRP1 inhibitors may increase the LGR5+ cell population, but could potentially prevent cell differentiation as these cells move out of the Matrix to form the hair shaft. The only potentially safe and effective way to upregulate Wnt only in the niches where it should be upregulated seems to be R-spondin. This presents its own difficulties with procurement and application, and safety is not assured even though in minimal mouse testing it did not induce tumor formation. ZNRF3/RNF43 inhibition would upregulate Wnt also, but inhibition of those two genes causes tumor formation within 3 weeks in mice. So we are back to broad-based GSK3b inhibition.
The combination of CHIR9902 and valproic acid has been shown in intestinal stem cells to be extremely efficient at increasing the number LGR5+ cells.
The caveats here are:
1. This combination promotes self-renewal of stem cells, but prevents their differentiation. The cells have to be removed from the CV culture before they can differentiate.
2. Safety is again a question mark.
3. R-spondin and Shh are still required. "R-spondin 1 and Noggin[upregulated by Shh] were still required to maintain the self-renewal of Lgr5+ stem cells in CV conditions."
4. The potential for stem cell depletion
The use of an mTOR inhibitor like rapamycin may theoretically prevent stem cell depletion. From the study: "Increasing the concentration of CHIR partially eliminated the need for R-spondin 1". Perhaps the levels of r-spondin in bald scalp are sufficient when combined with these drugs? CHIR+VPA for a limited time might renew the population of LGR5+ cells. If they do indeed block differentiation in vivo, removing them after 2 months while continuing to use a mild Wnt agonist and an AR antagonist might allow them to remain and to differentiate. It probably wouldn't work, but I am tempted to try this combination for two months with halcinonide, pyrilutamide, minoxidil, and ASC-J9, and after two months continue the protocol without CHIR/VPA. I doubt that I will try this because of the risk of cancer. This combination might be fine locally, but if it goes systemic it seems the combination of CHIR/VPA alone would cause cancer. LGR5+ cells are the culprit for initiating colon cancer, and implicated in other cancers as well. Imagine increasing them 100 fold throughout your whole body.
KRT15(hi) stem cells were maintained in bald scalp samples. However, CD200(hi)ITGA6(hi) and CD34(hi) cell populations--which both possessed a progenitor phenotype, in that they localized closely to the stem cell-rich bulge area but were larger and more proliferative than the KRT15(hi) stem cells--were markedly diminished
Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells - PMC
Androgenetic alopecia (Androgenetic Alopecia), also known as common baldness, is characterized by a marked decrease in hair follicle size, which could be related to the loss of hair follicle stem or progenitor cells. To test this hypothesis, we analyzed bald and ...
There's a lack of CD34+ and LGR5+ cells in bald scalp. After injury, an inflammatory response, involving NF-KB and Myc, causes CD34+ cells to divide. In the presence of Wnt signaling these and other stem cells are converted to LGR5+ cells. CD34+ cells are not required for hair growth. Their deletion has been shown not to prevent hair growth, but LGR5 is required for hair growth.
Lgr5+ cells are indispensable for telogen-to-anagen transition and that recovery of Lgr5+ cells is required for hair regeneration
the generation of Lgr5+ cells in the lower bulge and the hair germ from CD34+ stem cells is absolutely dependent on Wnt signalling
the Wnt pathway controls the recovery of Lgr5+ cells and inhibition of Wnt signalling prevents Lgr5+ cell and hair germ recovery.
It's evident that Wnt target genes are required for hair growth and are downregulated in bald scalp.
In fact, a new study finds that the only signals required for hair growth are Shh and Wnt.
When the genes for the sonic hedgehog and WNT signals were overexpressed, it was found that dermal condensates would form spontaneously without the need for other signals along the cell differentiation pathway. This demonstrated that these two signals are sufficient for the formulation of hair follicles. The researchers also found that the sonic hedgehog and WNT signals work hand-in-hand. They are codependent and work together in a very specific manner.
“We were able to see the dermal condensate without the existence of the other external signals, so that proves that not only are these signals [sonic hedgehog and WNT] essential, but also sufficient in dermal condensate formation,” explained Qu.

Decomposing a deterministic path to mesenchymal niche formation by two intersecting morphogen gradients
Organ formation requires integrating signals to coordinate proliferation, specify cell fates, and shape tissue. Tracing these events and signals remai…

Yale researchers find the signals essential for hair follicle formation
Researchers at the Yale School of Medicine found two signals that are essential and sufficient to a cell's differentiation into a dermal condensate.
Unfortunately both of these pathways are involved in tumorigenesis. The highly potent Smoothened(Shh) agonists, SAG and purmorphamine are too risky to use. There is a group of glucocorticoids that have been shown to potently induce Shh, and have been used regularly without causing cancer. Halcinonide is the most potent. That leaves Wnt activation. What compounds can be used for that? Direct Wnt receptor agonists are out of the question. Exogenous Wnt proteins have been shown to cause cancer in mice, just like SAG. In limited testing R-spondin did not have this effect, and it upregulates Wnt signaling only where it is supposed to be upregulated.
Proper lineage differentiation and crypt-villus organization within small intestinal organoids relies on an interesting property of R-spondin1. Namely, it augments preexisting domains of Wnt signaling in the crypt bottom (68) rather than inducing Wnt signaling de novo. Thus, when cells exit the crypt bottom–like structures of mini-guts and the spatial reach of Wnt, intestinal differentiation occurs normally (76), accounting for proper organoid architecture. In contrast, spatially uniform Wnt activation by GSK3 inhibition captures a rather homogeneous population of Lgr5+ stem cells in vitro in the absence of differentiated lineages
Uniformly upregulating Wnt signaling using GSK3b inhibitors or SFRP1 inhibitors may increase the LGR5+ cell population, but could potentially prevent cell differentiation as these cells move out of the Matrix to form the hair shaft. The only potentially safe and effective way to upregulate Wnt only in the niches where it should be upregulated seems to be R-spondin. This presents its own difficulties with procurement and application, and safety is not assured even though in minimal mouse testing it did not induce tumor formation. ZNRF3/RNF43 inhibition would upregulate Wnt also, but inhibition of those two genes causes tumor formation within 3 weeks in mice. So we are back to broad-based GSK3b inhibition.
The combination of CHIR9902 and valproic acid has been shown in intestinal stem cells to be extremely efficient at increasing the number LGR5+ cells.
The caveats here are:
1. This combination promotes self-renewal of stem cells, but prevents their differentiation. The cells have to be removed from the CV culture before they can differentiate.
2. Safety is again a question mark.
3. R-spondin and Shh are still required. "R-spondin 1 and Noggin[upregulated by Shh] were still required to maintain the self-renewal of Lgr5+ stem cells in CV conditions."
4. The potential for stem cell depletion
The use of an mTOR inhibitor like rapamycin may theoretically prevent stem cell depletion. From the study: "Increasing the concentration of CHIR partially eliminated the need for R-spondin 1". Perhaps the levels of r-spondin in bald scalp are sufficient when combined with these drugs? CHIR+VPA for a limited time might renew the population of LGR5+ cells. If they do indeed block differentiation in vivo, removing them after 2 months while continuing to use a mild Wnt agonist and an AR antagonist might allow them to remain and to differentiate. It probably wouldn't work, but I am tempted to try this combination for two months with halcinonide, pyrilutamide, minoxidil, and ASC-J9, and after two months continue the protocol without CHIR/VPA. I doubt that I will try this because of the risk of cancer. This combination might be fine locally, but if it goes systemic it seems the combination of CHIR/VPA alone would cause cancer. LGR5+ cells are the culprit for initiating colon cancer, and implicated in other cancers as well. Imagine increasing them 100 fold throughout your whole body.

