- Reaction score
- 3,026
As far as I know, I believe I have read all the studies on RU58841. There are not that many of them. Although many people worry about the stability of RU58841 in solution, from what I have read, there is no published evidence suggesting this should be a concern. I am not sure how this "rumor" began, but I think it can just be called that at this stage.
The best "evidence" I have seen in trying to investigate this has come from this site, which is a competitor of Kane's. It suggests that since nilutamide is supposedly prone to hydrolysis (breakdown in the presence of water), and nilutamide is similar to RU58841 in structure, so too must be RU58841.
Absolutely, nilutamide is quite similar to RU58841.
This is RU58841:
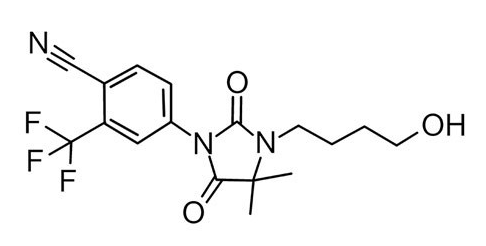
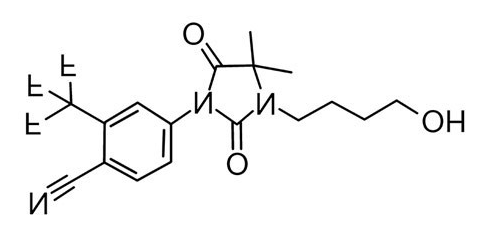
Or flipped upside down for better comparison it looks like this:
This is nilutamide:
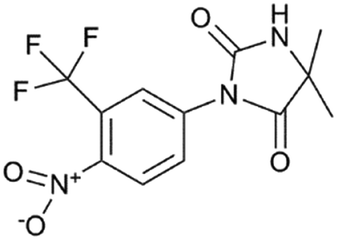
Once you flip the RU so they're oriented the same way, I don't think you have to be an expert in chemistry to see the similarities. Most androgens receptor antagonists have similarities in structure, and these two are more similar than most.
So perhaps the stability of RU58841 could be inferred from the stability of nilutamide. However, the claim RU58841.info makes about nilutamide being prone to breakdown in water is absolutely false.
I have reviewed the article they reference that they claim suggests this. I have attached it for posterity. So far as I can tell, the article actually makes no such claim.
To the contrary, here is what the article actually says, which is the same thing twice:
Abstract: Nilutamide showed significant degradation under basic hydrolysis and photolytic stress conditions, while it was stable to neutral, acidic and thermal stress conditions.
Discussion: The drug forms four degradation products, DP-I (retention time, Rt = 7.9 min), DP-II (Rt = 5.6 min), DP-III (Rt = 5.8 min) and DP-IV (Rt = 8.4) min under alkaline hydrolysis, and fifth, DP-V (Rt = 8.8 min), under photolytic conditions, while it was stable to neutral, acidic and thermal stress conditions.
"Neutral" stress conditions meant exposing it to neutral pH water for 48 hours. Acidic conditions meant adding acid, alkaline conditions meant adding base. Photolytic stress was stress from strong UV light. Thermal stress was heating it.
Out of all that, only under alkaline conditions in the presence of water did nilutamide significantly break down. There was a mild degree of breakdown under photolytic stress. This is why Kane ships in dark bottles - to protect from light, and you should keep it in dark bottles. But there was no breakdown under neutral water exposure, acidic water exposure, or heat stress conditions.
Even with light, the risk of breakdown is very low. You can see the degree of minimal breakdown under UV stress here:
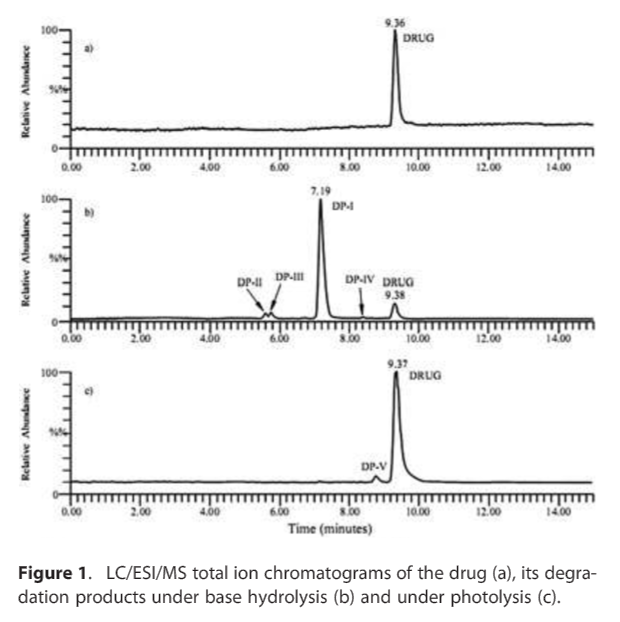
The "photolysis" breakdown product is labelled DP-V on the bottom diagram, and as you can see it's just a tiny bump, indicating even under hard UV stress it barely broke down at all.
Solution stability tests in the actual kinds of solvents we use for topical hair loss treatment (ie. PG-based solutions) are very hard to run as describe here, so it would be difficult to objectively confirm that RU58841 is stable in a practical real world solution.
Kane has claimed RU he has tested after leaving it in ethanol (which will have some water in it) for 6 months likely in the dark has been stable. I see no reason to disbelieve him. Since there will always be a bit of light exposure, we shouldn't expect such absolutely perfect long term perfection in real life. But no one is putting their bottles of RU in a tanning bed either.
In my opinion, this is therefore likely just an unfounded urban legend, and probably RU is stable in neutral pH solutions like we generally use, as long as you keep it in a dark bottle and out of direct sunlight.
That said, in my opinion, there isn't much benefit in mixing more than a few weeks worth of any topical solution at a time. It's not that much labor to mix these, and it will give you peace of mind to do them more frequently. When I first started with RU, I mixed daily, but in the past year, I have generally mixed ~10 days at a time, which I find is a reasonable compromise.
I think you could certainly try to do bigger and longer batches if you felt inclined. At least there doesn't seem to be any evidence to say you shouldn't.
The best "evidence" I have seen in trying to investigate this has come from this site, which is a competitor of Kane's. It suggests that since nilutamide is supposedly prone to hydrolysis (breakdown in the presence of water), and nilutamide is similar to RU58841 in structure, so too must be RU58841.
Absolutely, nilutamide is quite similar to RU58841.
This is RU58841:
Or flipped upside down for better comparison it looks like this:
This is nilutamide:
Once you flip the RU so they're oriented the same way, I don't think you have to be an expert in chemistry to see the similarities. Most androgens receptor antagonists have similarities in structure, and these two are more similar than most.
So perhaps the stability of RU58841 could be inferred from the stability of nilutamide. However, the claim RU58841.info makes about nilutamide being prone to breakdown in water is absolutely false.
I have reviewed the article they reference that they claim suggests this. I have attached it for posterity. So far as I can tell, the article actually makes no such claim.
To the contrary, here is what the article actually says, which is the same thing twice:
Abstract: Nilutamide showed significant degradation under basic hydrolysis and photolytic stress conditions, while it was stable to neutral, acidic and thermal stress conditions.
Discussion: The drug forms four degradation products, DP-I (retention time, Rt = 7.9 min), DP-II (Rt = 5.6 min), DP-III (Rt = 5.8 min) and DP-IV (Rt = 8.4) min under alkaline hydrolysis, and fifth, DP-V (Rt = 8.8 min), under photolytic conditions, while it was stable to neutral, acidic and thermal stress conditions.
"Neutral" stress conditions meant exposing it to neutral pH water for 48 hours. Acidic conditions meant adding acid, alkaline conditions meant adding base. Photolytic stress was stress from strong UV light. Thermal stress was heating it.
Out of all that, only under alkaline conditions in the presence of water did nilutamide significantly break down. There was a mild degree of breakdown under photolytic stress. This is why Kane ships in dark bottles - to protect from light, and you should keep it in dark bottles. But there was no breakdown under neutral water exposure, acidic water exposure, or heat stress conditions.
Even with light, the risk of breakdown is very low. You can see the degree of minimal breakdown under UV stress here:
The "photolysis" breakdown product is labelled DP-V on the bottom diagram, and as you can see it's just a tiny bump, indicating even under hard UV stress it barely broke down at all.
Solution stability tests in the actual kinds of solvents we use for topical hair loss treatment (ie. PG-based solutions) are very hard to run as describe here, so it would be difficult to objectively confirm that RU58841 is stable in a practical real world solution.
Kane has claimed RU he has tested after leaving it in ethanol (which will have some water in it) for 6 months likely in the dark has been stable. I see no reason to disbelieve him. Since there will always be a bit of light exposure, we shouldn't expect such absolutely perfect long term perfection in real life. But no one is putting their bottles of RU in a tanning bed either.
In my opinion, this is therefore likely just an unfounded urban legend, and probably RU is stable in neutral pH solutions like we generally use, as long as you keep it in a dark bottle and out of direct sunlight.
That said, in my opinion, there isn't much benefit in mixing more than a few weeks worth of any topical solution at a time. It's not that much labor to mix these, and it will give you peace of mind to do them more frequently. When I first started with RU, I mixed daily, but in the past year, I have generally mixed ~10 days at a time, which I find is a reasonable compromise.
I think you could certainly try to do bigger and longer batches if you felt inclined. At least there doesn't seem to be any evidence to say you shouldn't.
Attachments
Last edited:
