I looked into DNMT1 inhibitors some time ago to demethylate HFSCs, but ultimately I did not try it due to this study showing epidermal deletion of DNMT1 induces severe hair loss in mice, and this one showing that increased methylation of the AR in occipital HF may protect them from balding. However, I came across this study on reproductive follicles that made me revisit the idea.
DNA methylation mediated RSPO2 to promote follicular development in mammals
Furthermore:
RSPO in two studies is the second most highly correlated gene with Androgenetic Alopecia after the androgen receptor. Its induction has been shown to induce precocious telogen-anagen transition and to prevent catagen, so anything that upregulates RSPO is worth close inspection.
The following studies explore the use of DNMT1 inhibitors in cochlea hair follicle regeneration:
The DNA methylation inhibitor RG108 protects against noise-induced hearing loss
This leads us to a patent filed by George Cotsarelis for the use of a DNMT inhibitor for WIHN. When applied topically for 2 consecutive days 5-aza-2’-deoxycytidine increased hair follicle generation by 8-fold. There was nothing else used, no minoxidil, just two applications of this DNMT1 inhibitor.
Curiously, systemic administration of the compound had the opposite effect, and applying the topical compound for 5 consecutive days instead of 2 reduced its effectiveness. This could be due to a temporal window during regenration after which DNMT1 inhibition must be stopped for the next step in regeneration to take place, or it could be due to cytotoxicity. If the latter is the case then RG108, which does not display cytotoxicity, may be better suited for this.
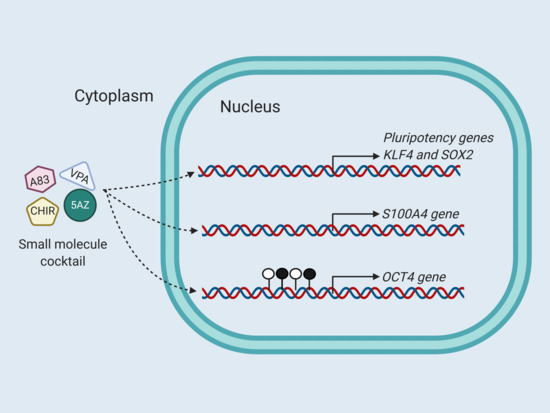
I believe this could complement the CHIR99021+VPA reprogramming experiment that some people are trying. This is all very serious stuff though that is risky to play around with. Cellular reprogramming is unpredictable, untested, and dangerous. We don't understand this stuff very well yet and the chance of causing cancer is real. This is all theoretical for now.
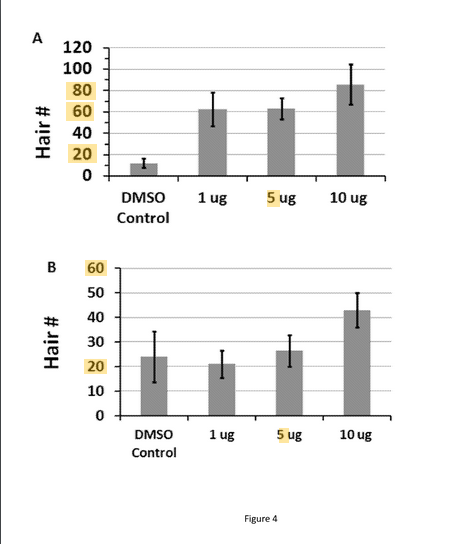
A. shows hair regeneration when applied for 2 days after scab detatchment. B. shows hair regeneration when applied for 5 days after scab detachment. This is even more effective than Cotsarelis' wounding study with SAG.
DNA methylation mediated RSPO2 to promote follicular development in mammals
RPSO2 is a master regulator of Wnt signaling, it primes stem cells for renewal and organ regeneration. When its expression is high the HF stays in anagenInhibition of DNA methylation or DNMT1 knockdown could decrease the methylation level of CpG island (CGI) in RSPO2 promoter and upregulate the expression level of RSPO2 in porcine GCs
CTNNB1 is B-catenin and E2 is 17b-estradiol.RSPO2 knockdown promoted the apoptosis of GCs, blocked the development of follicles, and delayed the onset of puberty with decreasing the expression level of Wnt signaling pathway-related genes (LGR4 and CTNNB1) in vivo. Taken together, the hypomethylation of −758/−749 and −563/−553 regions in RSPO2 promoter facilitated the occupancy of E2F1 and enhanced the transcription of RSPO2, which further promoted the proliferation and E2 secretion of GCs, inhibited the apoptosis of GCs, and ultimately ameliorated the development of follicles through Wnt signaling pathway. This study will provide useful information for further exploration on DNA-methylation-mediated RSPO2 pathway during follicular development.
Furthermore:
Of note here is that HSD17B1 is an estrogenic enzyme. It converts E1 into the more potent E2 and it also weakens androgenic activity by breaking down T into weaker androgens.To determine the effect of RSPO2 on the E2 secretion of GCs, the expression of genes related to estrogen signaling pathway were detected after overexpression and knockdown of RSPO2. qRT-PCR (Fig. 4A) and WB (Fig. 4B) results indicated that RSPO2 overexpression significantly promoted the mRNA and protein levels expression of CYP19A1 and HSD17B1, while RSPO2 silencing significantly inhibited the mRNA and protein expression levels of HSD17B1 as well as the protein expression level of CYP19A1. Moreover, RSPO2 overexpression significantly promoted the secretion of E2 in GCs, while RSPO2 knockdown displayed the opposite effects (Fig. 4C). Similarly, RSPO2-siRNA5 also significantly inhibited the mRNA expression of CYP19A1 and HSD17B1 (Fig. 4D) and the secretion of E2 in GCs. (Fig. 4E). In summary, RSPO2 promoted the secretion of E2 in porcine ovarian GCs.
Influence of estrogens on the androgen metabolism in different subunits of human hair follicles
RSPO in two studies is the second most highly correlated gene with Androgenetic Alopecia after the androgen receptor. Its induction has been shown to induce precocious telogen-anagen transition and to prevent catagen, so anything that upregulates RSPO is worth close inspection.
The following studies explore the use of DNMT1 inhibitors in cochlea hair follicle regeneration:
Epigenetic DNA Demethylation Causes Inner Ear Stem Cell Differentiation into Hair Cell-Like Cells
5-aza was applied to mouse utricle sensory epithelia-derived progenitor cells (MUCs) to investigate whether 5-aza stimulated MUCs to become sensory hair cells. After treatment, MUCs increased expression of hair cell genes and proteins. The DNA methylation level (indicated by percentage of 5-methylcytosine) showed a 28.57% decrease after treatment, which causes significantly repressed DNMT1 protein expression and DNMT activity. Additionally, FM1-43 permeation assays indicated that the permeability of 5-aza-treated MUCs was similar to that of sensory hair cells, which may result from mechanotransduction channels. This study not only demonstrates a possible epigenetic approach to induce tissue specific stem/progenitor cells to become sensory hair cell-like cells, but also provides a cell model to epigenetically modulate stem cell fate determination.
Generation of new hair cells by DNA methyltransferase (Dnmt) inhibitor 5-azacytidine in a chemically-deafened mouse model
This one shows that 5-aza upregulates SOX2 which is necessary for HF morphogenesis.Regeneration of mature mammalian inner ear hair cells remains to be a challenge. This study aims to evaluate the ability of DNA methyltransferase (Dnmt) inhibitor 5-azacytidine (5-aza) to generate outer hair cells (OHCs) in a chemically-deafened adult mouse model. 5-aza was administrated into the mouse inner ear via the round window. Immunofluorescence was used to examine the expression of hair cell specific proteins following 5-aza treatment. The results showed that in the chemically-deafened mouse cochlea, new OHCs were found post 5-aza treatment, whereas OHCs were completely lost in saline-treated mice. New hair cells expressed multiple hair cell markers included Myosin VIIa, Pou4f3 and Myosin VI. Newly-generated hair cells presented in three cochlear turns and were able to survive for at least six weeks. The effects of new hair cells generation by 5-aza were concentration dependent. Quantitative PCR study indicates that 5-aza may function through Dnmt1 inhibition. The results of this report suggest that the Dnmt inhibitor 5-aza may promote hair cell regeneration in a chemically-deafened mouse model.
The DNA methylation inhibitor RG108 protects against noise-induced hearing loss
This study suggests that inhibition of DNMT1 ameliorates noise-induced hearing loss and indicates that DNMT1 may be a promising therapeutic target.
Genome-Wide Demethylation by 5-aza-2’-Deoxycytidine Alters the Cell Fate of Stem/Progenitor Cells
DNA methyltransferase (DNMT) inhibitor 5-aza-2’-deoxycytidine (5-aza-CdR) is able to cause DNA demethylation in the genome and induce the expression of silenced genes. Whether DNA demethylation can affect the gene expression of stem/progenitor cells has not been understood. Mouse utricle epithelia-derived progenitor cells (MUCs), which possess stem cell features as previously described, exhibit a potential DNA methylation status in the genome. In this study, MUCs were treated with 5-aza-CdR to determine whether DNMT inhibitor is able to induce the differentiation of MUCs. With 5-aza-CdR treatment for 72 hr, MUCs expressed epithelial genes including Cdh1, Krt8, Krt18, and Dsp. Further, hair cell genes Myo7a and Myo6 increased their expressions in response to 5-aza-CdR treatment. The decrease in the global methylated DNA values after 5-aza-CdR treatment indicated a significant DNA demethylation in the genome of MUCs, which may contribute to remarkably increased expression of epithelial genes and hair cell genes. The progenitor MUCs then turned into an epithelial-like hair cell fate with the expression of both epithelial and hair cell genes. This study suggests that stem cell differentiation can be stimulated by DNA demethylation, which may open avenues for studying stem cell fate induction using epigenetic approaches.
This leads us to a patent filed by George Cotsarelis for the use of a DNMT inhibitor for WIHN. When applied topically for 2 consecutive days 5-aza-2’-deoxycytidine increased hair follicle generation by 8-fold. There was nothing else used, no minoxidil, just two applications of this DNMT1 inhibitor.
Currently, no therapy exists to regenerate normal skin that includes hair follicles and sweat glands from a wound or scar and hence, there is an existing need for such a therapy. [0003]The present invention addresses this need by providing DNA methyl transferase (DNMT) inhibitors, e.g., 5-aza-2′-deoxycytidine or 5-azacytidine. When inhibitors of DNMT activity (i.e., blocks DNA methylation) are administered following wound closure, enhanced hair follicle regeneration occurs, suggesting that DNA methyl transferase modulates the regeneration program in a strict temporal manner. Also demonstrated is that administration of 5-aza-2′-deoxycytidine or other related DNA methyltransferase inhibitors may promote skin regeneration in healing cutaneous wounds.
When applied to the wound site immediately following scab detachment for 2 days only, Aza-dC greatly enhanced hair follicle regeneration in healed wounds (from 3 to 8 fold) in a dose-dependent manner. (FIG. 4). Therefore, up-regulation of certain genes, via inhibition of DNA methylation, at the times when hair follicles begin to form plays a critical role in the hair follicle regeneration. This stimulatory effect on regeneration was diminished when the topical treatment was extended to 5 days (from SD0 to SD4), suggesting that the expression levels of some of these genes need to be temporally controlled to support hair follicle formation"
Curiously, systemic administration of the compound had the opposite effect, and applying the topical compound for 5 consecutive days instead of 2 reduced its effectiveness. This could be due to a temporal window during regenration after which DNMT1 inhibition must be stopped for the next step in regeneration to take place, or it could be due to cytotoxicity. If the latter is the case then RG108, which does not display cytotoxicity, may be better suited for this.
Topical Aza-dC treatment enhanced hair follicle regeneration in healed wounds. (A) 10 μL of Aza-dC (dissolved in DMSO) was applied topically to the wound site, once a day, for 2 consecutive days at the time of scab detachment (SD0 & SD1). Aza-dC treatment at all doses tested greatly enhanced hair follicle (HF) neogenesis compared to the vehicle control, with the strongest enhancement seen with the highest tested dose (10 μg). (A) 10 μL of Aza-dC (dissolved in DMSO) was applied topically to the wound site, once a day, for 5 consecutive days at the time of scab detachment (SD0 to SD1). While the two lower doses (1 & 5 μg) showed little effect on the regeneration, the highest dose (10 μg) still enhanced hair follicle regeneration.
I believe this could complement the CHIR99021+VPA reprogramming experiment that some people are trying. This is all very serious stuff though that is risky to play around with. Cellular reprogramming is unpredictable, untested, and dangerous. We don't understand this stuff very well yet and the chance of causing cancer is real. This is all theoretical for now.
A. shows hair regeneration when applied for 2 days after scab detatchment. B. shows hair regeneration when applied for 5 days after scab detachment. This is even more effective than Cotsarelis' wounding study with SAG.
Last edited: