Oh yeah, I am optimistic about this too, but I wish some one would just TRY IT ALREADY. It doesn't even have to be Christiano or Jahoda. It can be any doctor or dermatologist. A doctor from Yale named Brett King tried this in oral form on some one with AA and he regrew a full head of hair, after I believe more than 20 years of having no hair. There is no reason why a similar thing cannot be done with Androgenetic Alopecia in a topical form. And that is what frustrates a lot of us on here. All of this stuff gets tried for AA but never for Androgenetic Alopecia. I am sure it would not be hard at all for a doctor or researcher to find at least one person with Androgenetic Alopecia who has the financial means and the motivation to try this stuff in a topical form.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Dr. Angela Christiano - Targeting Immune Cells in Alopecia Areata
- Thread starter hellouser
- Start date
Yeah according to Christiano, they've taken it as far as they can without a partner stepping in. "All of us in academia are starving", she said. But it could be a physician prescribing topical Tof on an individual basis but Christiano herself, a researcher, can not because she doesn't have a license. There are already two JAK-I drugs approved now and several more in the pipeline. And with multiple label indications for JAK inhibitors in the offing in addition to rhematoid arthritis and blood cancer, like atopic dermatitis, vitiligo, psoriasis and clinical trials for AA, AU & AT already underway, it's not going to take a lot of convincing for a drug company to gamble on a couple of phase 1 trials to test against Androgenetic Alopecia. If someone took the lead right now and discovered in a short time that it works for Androgenetic Alopecia, 3 to 5 years to an approved drug to cure Androgenetic Alopecia. And she says this will be a "quick go-no go", meaning if it works for Androgenetic Alopecia like it does for AA, it will be obvious. What a great discovery it is.
- Reaction score
- 1,332
I find A.M Christiano her reasoning pretty weak. Based on observational evidence from people that have Androgenetic Alopecia and are on oral JAK inhibitors one would suspect that some changes would be seen. Just as case reports have come forward from people that take oral JAK inhibitors with AA. However no changes have been seen in people with Androgenetic Alopecia that are on oral JAK inhibitors.
So JAK inhibitors have shown to (sometimes) work orally for AA but not at all for Androgenetic Alopecia.
Now her reasoning is based on the rodent model where the rodents displayed faster anagen initiation upon topical application of JAK inhibitors. She argues that topical application might lead to increased concentrations of the drug in the hair follicle/skin and thus have a different effect.
This would have to mean that certain JAK inhibitors could have off target binding instead of only having biological activity towards the janus kinase enzymes at higher concentrations. It's possible but if I would have to bet my money on this I would immediately say that this is going to fail. Also again, using a rodent model and then translating it into humans for Androgenetic Alopecia is horrible. She knows this damn well.
Don't be fooled to compare AA with Androgenetic Alopecia too. There have been several case reports of AA reversal with different kinds of compounds. Honestly even spontaneous reversal without any intervention has been shown. Such observations are non-existent to Androgenetic Alopecia.
Don't get me wrong she has done a lot for hair loss related disorders. This is a great discovery for people that suffer from AA related conditions. But I fear that this is going to do sh*t all for Androgenetic Alopecia. What a dream it would be though . Miracles can happen.
. Miracles can happen.
So JAK inhibitors have shown to (sometimes) work orally for AA but not at all for Androgenetic Alopecia.
Now her reasoning is based on the rodent model where the rodents displayed faster anagen initiation upon topical application of JAK inhibitors. She argues that topical application might lead to increased concentrations of the drug in the hair follicle/skin and thus have a different effect.
This would have to mean that certain JAK inhibitors could have off target binding instead of only having biological activity towards the janus kinase enzymes at higher concentrations. It's possible but if I would have to bet my money on this I would immediately say that this is going to fail. Also again, using a rodent model and then translating it into humans for Androgenetic Alopecia is horrible. She knows this damn well.
Don't be fooled to compare AA with Androgenetic Alopecia too. There have been several case reports of AA reversal with different kinds of compounds. Honestly even spontaneous reversal without any intervention has been shown. Such observations are non-existent to Androgenetic Alopecia.
Don't get me wrong she has done a lot for hair loss related disorders. This is a great discovery for people that suffer from AA related conditions. But I fear that this is going to do sh*t all for Androgenetic Alopecia. What a dream it would be though
I find A.M Christiano her reasoning pretty weak. Based on observational evidence from people that have Androgenetic Alopecia and are on oral JAK inhibitors one would suspect that some changes would be seen. Just as case reports have come forward from people that take oral JAK inhibitors with AA. However no changes have been seen in people with Androgenetic Alopecia that are on oral JAK inhibitors.
How can we know which of the handful few test patients for rheumatoid arthritis or atopic dermatitis have Androgenetic Alopecia if any? By the same token, where are the doctors who knew their patients were both Androgenetic Alopecia and on the oral form of the drug for at least 8 months? Where are they, and why haven't they said, Uh-uh, this drug didn't do what you think it might do for Androgenetic Alopecia sufferers? All doctors and nurses are trained in the scientific method, and at least one would surely come forward with such information. So far nothing.
I can't see them even recording whether or not any of those patients are suffering from Androgenetic Alopecia because it would be irrelevant to those trial designs for specific immune disorders. Yes, other side effects such as hair growth would be reported, but how many of them are actually suffering from Androgenetic Alopecia who have been on Xeljanz, a drug with some awful side effects when taken orally, for a long enough time(8 months according to Brett King) to notice hair re-growth? And it's an expensive pill, too. Insurance companies won't be paying for a single independent/individual trial to test if it works for Androgenetic Alopecia we can be sure. Topical Tof is still in clinical trials for AA, vitiligo and atopic dermatitis apparently. Perhaps one of them will have Androgenetic Alopecia and report re-growth. It's still early days yet, and the number of people with Androgenetic Alopecia who have been given a topical dose of Tofacitinib?: zero. Only people with psoriasis and, by off chance, AA, have been prescribed Tof off-label and as an oral dose which is not as effective for re-growing hair as a topical delivery according to the few reports we do have. And after eight months of individual prescriptions to treat psoriasis etc, an even smaller subset of the already tiny super-set of people with immune disorders and who also happen to have AA, now have full heads of hair. It's an astonishing discovery given that prior to two years ago, solving AA was viewed as just as difficult a problem as Androgenetic Alopecia.
- Reaction score
- 1,332
How can we know which of the handful few test patients for rheumatoid arthritis or atopic dermatitis have Androgenetic Alopecia if any? By the same token, where are the doctors who knew their patients were both Androgenetic Alopecia and on the oral form of the drug for at least 8 months? Where are they, and why haven't they said, Uh-uh, this drug didn't do what you think it might do for Androgenetic Alopecia sufferers?
I can't see them even recording whether or not any of those patients are suffering from Androgenetic Alopecia because it would be irrelevant to those trial designs for specific immune disorders. Yes, other side effects such as hair growth would be reported, but how many of them are actually suffering from Androgenetic Alopecia who have been on Xeljanz, a drug with some awful side effects when taken orally, for a long enough time(8 months according to Brett King) to notice hair re-growth? And it's an expensive, too. Insurance companies won't be paying for a single independent/individual trial to test if it works for Androgenetic Alopecia we can be sure. Topical Tof is still in clinical trials for AA, vitiligo and atopic dermatitis apparently. Perhaps one of them will have Androgenetic Alopecia and report re-growth. It's still early days yet, and the number of people with Androgenetic Alopecia who have been given a topical dose of Tofacitinib?: 0.0 Only people with psoriasis and, by off chance, AA, have been guinea pigs for a topical, and after eight months of individual prescriptions to treat psoriasis etc, an even smaller subset of the already tiny superset of people with immune disorders and who also happen to have AA, now have full heads of hair. Amazing. Someone will surely test it for Androgenetic Alopecia after what has only been a relatively recent discovery that it re-grows hair for AA sufferers.
You say handful. Let's take look at Xeljanz;
The newly reported 12-month Oral Standard study included 717 patients with moderate-to-severe active RA who had previously responded inadequately to methotrexate therapy. Participants were randomized to receive either tofacitinib 5 mg or 10 mg twice a day
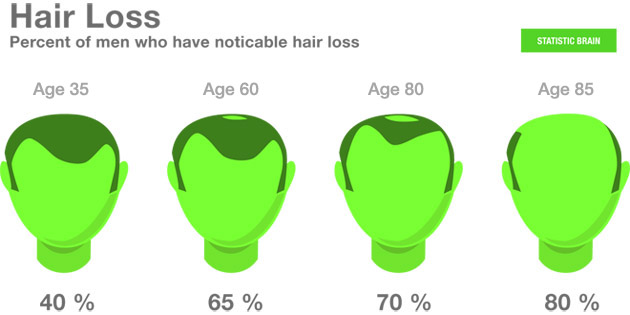
A 12 month study in 717 participants. Are you going to argue here that nobody of these 717 patients has Androgenetic Alopecia? Have you looked at the statistics of Androgenetic Alopecia? It's prevalent as hell I think you understand that;
Now this is ONE trial of ONE compound (Xeljanz). It has rune more lengthy phase 3 trials. Add to this the fact that this medicine is actually on the market now. Add to this also that multiple other JAK inhibitors are running clinical trials.
Hair regrowth would be noted easily if this was the case upon oral administration either by patient or observer. Case reports would already been flooding online. But this isn't the case. Thus we can already draw pretty much a conclusion that oral JAK inhibitors at the dosages described don't work for Androgenetic Alopecia. Hundreds of people have been evidence for this. This is how it works.
A.M Christiano understands this that is why she made the remark about possible increased concentrations of the drug by topical application. It's a weak argument in my opinion, but she has a point though. Drugs can show off target binding at increased concentrations. I guess we'll see. I don't think it's going to work for Androgenetic Alopecia at all nonetheless it's a great discovery for people suffering from AA related conditions.
That's the beauty of Androgenetic Alopecia, because it is so prevalent in people many are automatically a test subject in clinical trials unrelated to hair disorders. Nonetheless it's still important obviously they will test the stuff topically in Androgenetic Alopecia patients.
Yes as I said before, nurses and doctors are trained in the scientific method. They themselves could have noted the opposite - that test patients with Androgenetic Alopecia did not re-grow hair in the interest of science. So far none of those professional medical people have come forward and reported Tof failed to re-grow hair for patients with pattern baldness. And jumping to conclusions without actual data either way would be very unscientific.
You're talking about 717 test patients from Pfizer's clinical trial on Xeljanz for rheumatoid arthritis. And your diagram says a little more than half of males have some degree of Androgenetic Alopecia by middle age. It certainly suggests at least a few patients might have Androgenetic Alopecia but we still can't be sure. And we still don't know how many males with Androgenetic Alopecia were enrolled in the trials if any. Also, it's known that women suffer from rheumatoid arthritis at a rate three times that of men, so it might also be a safe bet that two-thirds of test patients are women without hair loss, but again, it's jumping to conclusions similarly. The point is, we don't know if any of the test patients were significantly bald from Androgenetic Alopecia. It certainly wouldn't have been a condition for enrollment in clinical trials. It's even possible there were none.
Furthermore, the young man with psoriasis (and Alopecia Areata) treated by Brett King was given a 15 mg oral dose of Tof for 8 months. All of Pfizer's test patients for rheumatoid arthritis were given smaller doses of 5-10 mg plus 40 mg of adalimumab sub-cu every other week. That is a different protocol compared to what was given to psoriasis and AA test patients. Perhaps the handful few test patients who might possibly have both RA and Androgenetic Alopecia together were all put on lowest doses of Xeljanz.
Additionally, Christiano says delivery is where the opportunities lie for an Androgenetic Alopecia test protocol. She says it would be preferable that the drug remain in the dermis nearest follicles for longer than 5 minutes. Christiano-Jahoda method using 3d speroids for delivery of JAK inhibitors hasn't been tried on people yet let lone those with Androgenetic Alopecia. Short time to fail, she says. We should know sooner than later.
Finally, there are such things as confidentiality/disclosure agreements between companies and academia and doctors. It could even be a case that they, and perhaps through anecdotal and other evidence as you say should exist, already know the effects Xeljanz/Tofacitinib has on people with Androgenetic Alopecia but are restricted from publishing or broadcasting that information to the public. A lot of money and reputations at stake with a cure for Androgenetic Alopecia.
You're talking about 717 test patients from Pfizer's clinical trial on Xeljanz for rheumatoid arthritis. And your diagram says a little more than half of males have some degree of Androgenetic Alopecia by middle age. It certainly suggests at least a few patients might have Androgenetic Alopecia but we still can't be sure. And we still don't know how many males with Androgenetic Alopecia were enrolled in the trials if any. Also, it's known that women suffer from rheumatoid arthritis at a rate three times that of men, so it might also be a safe bet that two-thirds of test patients are women without hair loss, but again, it's jumping to conclusions similarly. The point is, we don't know if any of the test patients were significantly bald from Androgenetic Alopecia. It certainly wouldn't have been a condition for enrollment in clinical trials. It's even possible there were none.
Furthermore, the young man with psoriasis (and Alopecia Areata) treated by Brett King was given a 15 mg oral dose of Tof for 8 months. All of Pfizer's test patients for rheumatoid arthritis were given smaller doses of 5-10 mg plus 40 mg of adalimumab sub-cu every other week. That is a different protocol compared to what was given to psoriasis and AA test patients. Perhaps the handful few test patients who might possibly have both RA and Androgenetic Alopecia together were all put on lowest doses of Xeljanz.
Additionally, Christiano says delivery is where the opportunities lie for an Androgenetic Alopecia test protocol. She says it would be preferable that the drug remain in the dermis nearest follicles for longer than 5 minutes. Christiano-Jahoda method using 3d speroids for delivery of JAK inhibitors hasn't been tried on people yet let lone those with Androgenetic Alopecia. Short time to fail, she says. We should know sooner than later.
Finally, there are such things as confidentiality/disclosure agreements between companies and academia and doctors. It could even be a case that they, and perhaps through anecdotal and other evidence as you say should exist, already know the effects Xeljanz/Tofacitinib has on people with Androgenetic Alopecia but are restricted from publishing or broadcasting that information to the public. A lot of money and reputations at stake with a cure for Androgenetic Alopecia.
- Reaction score
- 1,332
I'm not talking only about these 717 patients. I said this;
I'm talking about hundreds of patients more if not thousand of patients. Yes not everyone of them will have Androgenetic Alopecia, not everyone of them will be obviously men. However a pretty big % will have Androgenetic Alopecia to some degree.These professional medical people don't have to argue that tofacitinib hasn't failed orally at the dosages tested for Androgenetic Alopecia, because it has. Do you want me to actually contact A.M Christiano and see if she wants to argue otherwise vs me? I can do that if you want. Would that convince you?
Hair regrowth is something that doesn't go unnoticed dude. There is no difference between a norwood 2 and a norwood 6. The basic principle is the same. A norwood 2 wants to have reversal of miniaturization in the norwood 2 area and the norwood 6 wants to have it in the norwood 6 area.
Already in the trial I mentioned we see these dosages. Actually in the trial one group had 2x 10mg which amounts to 20mg a day, which is even a bigger dosage than in the AA guy. Here is another phase 3 trial of tofacitinib;
And here is another one for psoriasis oral for a 1 year period https://www.aad.org/eposters/Submissions/getFile.aspx?id=2216&type=sub. With N= 745 and N= 741 on 10mg and 20mg daily, almost 1500 patients.
If we only count these trials I just mentioned this is already over 2500 people. Add to this actually more trials that have run, add to this other JAK inhibitors that are in clinical trials and add to this that Xeljanz is actually launched on the market and in use by patients. We are talking about thousands of people here.
If you still argue that Xeljanz works orally for Androgenetic Alopecia at these dosages then you believe in fairy tales, don't you think?
Am I excluding here that perhaps Xeljanz won't work at even higher oral dosages or topically? No but clearly at these dosages it doesn't do sh*t at least orally. I also think the chances of it working in bigger dosages or topically is incredibly small, but we'll see. I have other reasoning behind this than only looking at the findings of Christiano. All she has is a rodent model which says nothing, nada. She wouldn't argue otherwise with this also. In fact she has actually mentioned recently herself that a rodent model doesn't translate to Androgenetic Alopecia.
Fine it's possible, weak argumentation imo. She has a point there. Like I said increased concentrations might have a other effect. I doubt it though. However again orally at these dosages described it's just not going to work. Where does she base her observations on though? That's right mice.
I think it's illegal to say otherwise. If hair growth would get noted either by patient or observer such findings need to be described in the clinical trial. I don't know this for sure though. Anyway they would be pretty damn happy to report such findings. Thinking that it would be kept secret is typical tinfoil hat thinking. It would be all over the news just as the AA guy. It's patented anyway.
All in all I wouldn't get your hopes up to much with this one. But time will tell. I hope I'm wrong obviously.
Now this is ONE trial of ONE compound (Xeljanz). It has rune more lengthy phase 3 trials. Add to this the fact that this medicine is actually on the market now. Add to this also that multiple other JAK inhibitors are running clinical trials
I'm talking about hundreds of patients more if not thousand of patients. Yes not everyone of them will have Androgenetic Alopecia, not everyone of them will be obviously men. However a pretty big % will have Androgenetic Alopecia to some degree.These professional medical people don't have to argue that tofacitinib hasn't failed orally at the dosages tested for Androgenetic Alopecia, because it has. Do you want me to actually contact A.M Christiano and see if she wants to argue otherwise vs me? I can do that if you want. Would that convince you?
Hair regrowth is something that doesn't go unnoticed dude. There is no difference between a norwood 2 and a norwood 6. The basic principle is the same. A norwood 2 wants to have reversal of miniaturization in the norwood 2 area and the norwood 6 wants to have it in the norwood 6 area.
After two months on tofacitinib at 10 mg daily, the patient’s psoriasis showed some improvement, and the man had grown scalp and facial hair — the first hair he’d grown there in seven years. After three more months of therapy at 15 mg daily
Already in the trial I mentioned we see these dosages. Actually in the trial one group had 2x 10mg which amounts to 20mg a day, which is even a bigger dosage than in the AA guy. Here is another phase 3 trial of tofacitinib;
The Oral Step study enrolled 399 patients with moderate-to-severe active RA who had previously demonstrated an inadequate response to at least one TNF inhibitor. Participants were randomized to receive 5 mg or 10 mg tofacitinib twice daily, or placebo, in addition to stable background methotrexate. All placebo-treated patients were switched over to tofacitinib therapy at the three-month mark.
And here is another one for psoriasis oral for a 1 year period https://www.aad.org/eposters/Submissions/getFile.aspx?id=2216&type=sub. With N= 745 and N= 741 on 10mg and 20mg daily, almost 1500 patients.
If we only count these trials I just mentioned this is already over 2500 people. Add to this actually more trials that have run, add to this other JAK inhibitors that are in clinical trials and add to this that Xeljanz is actually launched on the market and in use by patients. We are talking about thousands of people here.
If you still argue that Xeljanz works orally for Androgenetic Alopecia at these dosages then you believe in fairy tales, don't you think?
Am I excluding here that perhaps Xeljanz won't work at even higher oral dosages or topically? No but clearly at these dosages it doesn't do sh*t at least orally. I also think the chances of it working in bigger dosages or topically is incredibly small, but we'll see. I have other reasoning behind this than only looking at the findings of Christiano. All she has is a rodent model which says nothing, nada. She wouldn't argue otherwise with this also. In fact she has actually mentioned recently herself that a rodent model doesn't translate to Androgenetic Alopecia.
Additionally, Christiano says delivery is where the opportunities lie for an Androgenetic Alopecia test protocol. She says it would be preferable that the drug remain in the dermis nearest follicles for longer than 5 minutes. Christiano-Jahoda method using 3d speroids for delivery of JAK inhibitors hasn't been tried on people yet let lone those with Androgenetic Alopecia. Short time to fail, she says. We should know sooner than later.
Fine it's possible, weak argumentation imo. She has a point there. Like I said increased concentrations might have a other effect. I doubt it though. However again orally at these dosages described it's just not going to work. Where does she base her observations on though? That's right mice.
Finally, there are such things as confidentiality/disclosure agreements between companies and academia and doctors. It could even be a case that they, and perhaps through anecdotal and other evidence as you say should exist, already know the effects Xeljanz/Tofacitinib has on people with Androgenetic Alopecia but are restricted from publishing or broadcasting that information to the public. A lot of money and reputations at stake with a cure for Androgenetic Alopecia.
I think it's illegal to say otherwise. If hair growth would get noted either by patient or observer such findings need to be described in the clinical trial. I don't know this for sure though. Anyway they would be pretty damn happy to report such findings. Thinking that it would be kept secret is typical tinfoil hat thinking. It would be all over the news just as the AA guy. It's patented anyway.
All in all I wouldn't get your hopes up to much with this one. But time will tell. I hope I'm wrong obviously.
Swoop said:If you still argue that Xeljanz works orally for Androgenetic Alopecia at these dosages then you believe in fairy tales, don't you think?
No one is suggesting an oral dose for Androgenetic Alopecia. What scientists are saying is that a topical solution should be tried on patients with Androgenetic Alopecia because topical worked much better for growing hair in both lab tests and for AA patients. The only person I'm aware of who has tested topical Tof on patients with AA is Angela Christiano, and in her paper published just a few weeks ago she mentions a special delivery technique for Tof using 3-dimensional spheroids. And for some reason which no one really understands, it worked orally for Brett King's patient to re-grow all of his hair. However, Angela Christiano said they know from their own work that Tof works much better for growing hair in AA patients when applied topically and that it is doing something more than just stopping an auto-immune attack on hair follicles - apparently the drug affects several follicle growth pathways as well.
"The surprise was when we started using the drugs on alopecia areata patients, when we used them topically the hair grew back much faster and more robustly than it did orally," Christiano told NBC News.
Note that topical tofacitinib was not used in any of the clinical trials you've pointed out to us.
You could certainly try to contact Angela Christiano or Brett King. If so, they should explain to you why they think it would be worthwhile to do a pilot test for a topical version of Tofacitinib on people with pattern baldness, and why the real work left is with developing a delivery technique, and it will be a topical solution that is important to testing this drug's potential for hair growth for all of the major hair loss conditions. With actual phase 1 trials for Androgenetic Alopecia, ALL of the test patients will have it as a condition for enrollment. But guessing what percentage might have had Androgenetic Alopecia or why hair re-growth wasn't reported in clinical trials that had nothing to do with Androgenetic Alopecia, is not very scientific. If you do find out something more, please do get back to us.
- Reaction score
- 1,332
No one is suggesting an oral dose for Androgenetic Alopecia. What scientists are saying is that a topical solution should be tried on patients with Androgenetic Alopecia because topical worked much better for growing hair in both lab tests and for AA patients. The only person I'm aware of who has tested topical Tof on patients with AA is Angela Christiano, and in her paper published just a few weeks ago she mentions a special delivery technique for Tof using 3-dimensional spheroids. And for some reason which no one really understands, it worked orally for Brett King's patient to re-grow all of his hair. However, Angela Christiano said they know from their own work that Tof works much better for growing hair in AA patients when applied topically and that it is doing something more than just stopping an auto-immune attack on hair follicles - apparently the drug affects several follicle growth pathways as well.
Note that topical tofacitinib was not used in any of the clinical trials you've pointed out to us.
You could certainly try to contact Angela Christiano or Brett King. If so, they should explain to you why they think it would be worthwhile to do a pilot test for a topical version of Tofacitinib on people with pattern baldness, and why the real work left is with developing a delivery technique, and it will be a topical solution that is important to testing this drug's potential for hair growth for all of the major hair loss conditions. With actual phase 1 trials for Androgenetic Alopecia, ALL of the test patients will have it as a condition for enrollment. But guessing what percentage might have had Androgenetic Alopecia or why hair re-growth wasn't reported in clinical trials that had nothing to do with Androgenetic Alopecia, is not very scientific. If you do find out something more, please do get back to us.
Never did I say otherwise. I just said the evidence is goddamn weak. Ultimately the only evidence she has is that it causes hair growth in mice and remember these mice only displayed a faster and more robust onset of hair growth when applied topically instead of systemically. They did get hair regrowth when the compound was given systemically. So if we look at the observations in the mice why don't we draw a line of thinking that at least a oral formulation in humans at X dosage at least has to display some sort of hair growth in Androgenetic Alopecia patients?
It's the same thing over and over again dude. There is zero I repeat zero evidence of anything occurring in humans with JAK inhibitors thus far. I'm not saying it's not possible that a topical formulation won't work, but I don't see anything exciting about this at all.
According to Christiano, tufts of hair and spotty not remarkable growth as observed with topical delivery. The only way to know for sure is to test topical delivery(and Christiano's & Jahoda's method for topical delivery would be nice) on people with Androgenetic Alopecia and using the scientific method, i.e. double blind, using a range of doses and every test subject diagnosed with Androgenetic Alopecia.
Progress in science and tech comes from doing things differently in unpredictable ways. In science and tech there is a history of challenges, made by people who knew little about the task at hand, against what were "impossible" claims. A lead Royal Astronomer once scoffed at the idea of trans-oceanic voyages saying heavier than water ships would sink to the bottom. He said we might as well try flying to the moon.
- Reaction score
- 1,332
According to Christiano, tufts of hair and spotty not remarkable growth as observed with topical delivery. The only way to know for sure is to test topical delivery(and Christiano's & Jahoda's method for topical delivery would be nice) on people with Androgenetic Alopecia and using the scientific method, i.e. double blind, using a range of doses and every test subject diagnosed with Androgenetic Alopecia.
Progress in science and tech comes from doing things differently in unpredictable ways. In science and tech there is a history of challenges, made by people who knew little about the task at hand, against what were "impossible" claims. A lead Royal Astronomer once scoffed at the idea of trans-oceanic voyages saying heavier than water ships would sink to the bottom. He said we might as well try flying to the moon.
I don't find this particular exciting research or doing things differently. It all boils down principally to observations made in the rodent model. I don't get excited from a rodent model because it has failed time and time again, you seem to be way more excited about that. For what reason really? I have spoken to some (elite) researchers and they all concur that the rodent model is bad in terms of translating drug testing to Androgenetic Alopecia. Everyone can see this, right?
Had the research be done on some sort of improved model or had there been any evidence of hair regrowth occurring in somebody with Androgenetic Alopecia then my stance would have been different. But this is clearly not the case. I often question the media publicity and such "hype" to research like this. Is it really because the compound has a high chance of achieving something for Androgenetic Alopecia or is such media attention rather for personal benefit?
This is what actual scientists and experts in hair biology are excited about in addition to the fact that they are on the verge of solving alopecia areata, alopecia, totalis and universalis. Until recently those hair loss disorders were considered brutally hard problems for science to solve. I would say just as if not more difficult because of the extent of the hair loss. Imagine losing all of your hair even in the occipital region. Imagine having zero healthy hair follicles for transplantation let alone a combover a la Donald Trump.
JAK inhibitors stimulate hair follicles out of the resting phase into the growing phase. They also show the ability to elongate the growing phase.
JAK inhibitors activate the Wnt and Shh signaling pathway.
JAK inhibitors cause the activation of hair follicle progenitor (stem) cells.
Tofacitinib (a JAK inhibitor) promotes inductivity of dermal papillae cells.
Tofacitinib treatment promotes growth of human Hair Follicles
Hair-inducing effects of JAK inhibition are not dependent on the activity of lymphocytes
Tofacitinib promotes hair growth by positively affecting genes within pathways that are crucial to hair growth. For example: TGFB pathway genes, BMP pathway genes, LEF1, and NOTCH pathway genes.
As well, the scientific study also shows results from using a “Sonic Hedgehog” pathway agonist although not as impressive as the results for JAK inhibition. Three down one to go.
- Reaction score
- 1,332
This is what they are excited about in addition to the fact that they are on the verge of solving alopecia areata, alopecia, totalis and universalis. Until recently those hair loss disorders were considered brutally hard problems for science to solve. I would say just as if not more difficult because of the extent of the hair loss. Imagine losing all of your hair even in the occipital region. Imagine having zero healthy hair follicles for transplantation let alone a combover a la Donald Trump.
As well, the scientific study also shows results from using a “Sonic Hedgehog” pathway agonist although not as impressive as the results for JAK inhibition. Three down one to go. :salut:
Comparing AA with Androgenetic Alopecia is comparing apples with pears. AA is a autoimmune disease that is driven by cytotoxic T cells. It is simplistic as hell compared to Androgenetic Alopecia.
Androgenetic Alopecia is on a whole different level and isn't a autoimmune disease. Absolutely great that JAK inhibitors seem to be more effective than other treatments for AA. Although many compounds have shown to be effective in AA to some extent (mainly immunomodulatory compounds just as JAK inhibitors) and honestly even spontaneous reversal sometimes occurs with AA. Such observations again like I mentioned are non-existent to Androgenetic Alopecia.
It would be absolutely great that this is going to work for Androgenetic Alopecia but again the evidence is extremely weak, the only thing you can come up with is that it spurs hair growth in mice.
If we had to rely on mice results or in vitro HF explant models we would have been cured 100 times already. Doesn't work like that. I guess we'll see soon, time will tell.
It's time researchers actually start developing better models for drug testing
HairlossCurse
Member
- Reaction score
- 39
Wow, didn't realize how big this was until I read the paper. The jak inhibitors promote the onset of anagen (growth phase) by reactivating the progenitor cells. Many non scarring types of alopecias (eg aa and Androgenetic Alopecia) seem to keep hair follicle stem cells but lose progenitor cells. This could be a way to reactivate the progenitor cells.
I saw an important point:
"These two findings are reconciled when considering hair growth in AA patients as a two-step mechanism: first, the T cell–mediated immune attack on epithelial cells must be eliminated, and second, anagen growth must be reinitiated "
It was just a bit of luck that, in AA, this drug was able to do both at once.
I believe PGD2 plays the role of the bad guy that must be knocked out in Androgenetic Alopecia.
PGD2 promotes catagen(dormant phase) and increases catagen phase. It says in the study that Jak inhibitors are effective during the telegen phase. I don't think jak inhibitors will be able to bypass the effect of pgd2 completely, but if we nullify the effect of pgd2, just like the T-cell thing was nullified. I think we are in business.
My theory:
In AA,
1. Nullify the T cell–mediated immune attack on epithelial cells (solution: JAK inhibitors)
2. Restart anagen (reactivate progenitor cells) solution: JAK inhibitors
In Androgenetic Alopecia,
1. Nullify the effect of PGD2 (solution: setipiprant)
2. Restart anagen (reactivate progenitor cells) solution: JAK inhibitors
I saw an important point:
"These two findings are reconciled when considering hair growth in AA patients as a two-step mechanism: first, the T cell–mediated immune attack on epithelial cells must be eliminated, and second, anagen growth must be reinitiated "
It was just a bit of luck that, in AA, this drug was able to do both at once.
I believe PGD2 plays the role of the bad guy that must be knocked out in Androgenetic Alopecia.
PGD2 promotes catagen(dormant phase) and increases catagen phase. It says in the study that Jak inhibitors are effective during the telegen phase. I don't think jak inhibitors will be able to bypass the effect of pgd2 completely, but if we nullify the effect of pgd2, just like the T-cell thing was nullified. I think we are in business.
My theory:
In AA,
1. Nullify the T cell–mediated immune attack on epithelial cells (solution: JAK inhibitors)
2. Restart anagen (reactivate progenitor cells) solution: JAK inhibitors
In Androgenetic Alopecia,
1. Nullify the effect of PGD2 (solution: setipiprant)
2. Restart anagen (reactivate progenitor cells) solution: JAK inhibitors
Comparing AA with Androgenetic Alopecia is comparing apples with pears. AA is a autoimmune disease that is driven by cytotoxic T cells. It is simplistic as hell compared to Androgenetic Alopecia.
I'll agree with you on two points: 1. Mouse models are a poor approximation for human hair biology and 2. We won't know until someone actually tries it
Progress in science and technology comes from doing things differently in unpredictable ways. And this is exactly what Christiano et al have done in discovering the wonderful hair growth properties of JAK inhibitors. The fact that 3 or 4 AA, AT and AU patients have re-grown all their hair should be viewed as breathtakingly good news for anyone with hair loss.
And thank goodness we have people in the doing department otherwise it wouldn't matter which one of the three major hair loss disorders remains unsolved, and we'd be stuck with "the big three" for all time.
- Reaction score
- 1,332
I'll agree with you on two points: 1. Mouse models are a poor approximation for human hair biology and 2. We won't know until someone actually tries it
Progress in science and technology comes from doing things differently in unpredictable ways. And this is exactly what Christiano et al have done in discovering the wonderful hair growth properties of JAK inhibitors. The fact that 3 or 4 AA, AT and AU patients have re-grown all their hair should be viewed as breathtakingly good news for anyone with hair loss.
And thank goodness we have people in the doing department otherwise it wouldn't matter which one of the three major hair loss types remains unsolved, and we'd be stuck with "the big three" for all time.
It is absolutely amazing indeed for people suffering from these hair loss related disorders, I agree. Here this guy just posted here this week; http://www.hairlosstalk.com/interact/showthread.php/95913-My-Story-17-years-old. He can have a positive outlook because it's very likely he will get all his hair back. Absolutely awesome, I am happy for these people. JAK inhibitors seem to do well for a wide arrange of auto immune disorders actually and the news hype for AA was totally justified. Why? Cause it was displayed in humans, not in mice.
Ultimately I think drugs will probably not be our answer to Androgenetic Alopecia anyway as a cure. Luckily we got rapid advancements in regenerative therapy going (will also deal with the androgen sensitivity). Keep an eye on the likes of Takashi Tsuji!
Luckily we got rapid advancements in regenerative therapy going (will also deal with the androgen sensitivity). Keep an eye on the likes of Takashi Tsuji!
Thanks good post!
- - - Updated - - -
I saw an important point:
"These two findings are reconciled when considering hair growth in AA patients as a two-step mechanism: first, the T cell–mediated immune attack on epithelial cells must be eliminated, and second, anagen growth must be reinitiated "
It was just a bit of luck that, in AA, this drug was able to do both at once.
Good post! Yes, what you said about combining a JAK-I with a PGD2 inhibitor could be what everyone's looking for. Christiano mentioned Kythera and setipiprant recently and said it sounds encouraging. I have a strong feeling that news announcements about hair loss research and treatments will be stepped up to a frenzied pace in the next few years. JAK-I plus setipiprant sounds like it could be the dynamic duo for hair loss. You are all very knowledgeable about hair loss here. Great posts both of you.
I'll agree with you on two points: 1. Mouse models are a poor approximation for human hair biology and 2. We won't know until someone actually tries it
Progress in science and technology comes from doing things differently in unpredictable ways. And this is exactly what Christiano et al have done in discovering the wonderful hair growth properties of JAK inhibitors. The fact that 3 or 4 AA, AT and AU patients have re-grown all their hair should be viewed as breathtakingly good news for anyone with hair loss.
And thank goodness we have people in the doing department otherwise it wouldn't matter which one of the three major hair loss disorders remains unsolved, and we'd be stuck with "the big three" for all time.
That is the key phrase: "we won't know until someone actually tries it". And as far as Androgenetic Alopecia, there is no evidence that someone is actually trying it. Sure it's great that this is working for people with AA, but lots of things can regrow hair in people with AA. For instance, Angela Christiano had AA and actually regrew her hair with cortisone injections I believe. Another guy's AA was cured with PRP, which so far hasn't done anything for Androgenetic Alopecia. At least I've actually seen people with AA recover and grow all their hair back. I've never seen that with Androgenetic Alopecia. But you said it NewUser, "we won't know until someone actually tries it".
Sure it's great that this is working for people with AA, but lots of things can regrow hair in people with AA. For instance, Angela Christiano had AA and actually regrew her hair with cortisone injections I believe.
She said her hair is thinner than before, and that she has an eyebrow that comes and goes. I don't believe they've seen any drug push hair follicles into anagen growth before like topical Tof. Apparently this discovery blew their socks off so to speak.

