Histogen Signs Deal for Commercialization of HSC in China
by Kevin Rands | September 29, 2016 11:47 am
Previously we wrote about Histogens “Hair Stimulating Complex” research project which was received with quite a bit of curiosity due to the notable before and after photos provided by the research team. One of those photos that we were provided can be seen in the header of this article. In our article back in August: Histogen – Hair Stimulating Complex Research Update[1], Dr. Gail Naughton announced their hopes to finalize an agreement with a company in China for the development and commercialization of Hair Stimulating Complex (HSC).
Today they’ve announced that this agreement has been finalized. This opens the way for Histogen and their partner in China – Pineworld Capital, Ltd to begin laying out a plan for human clinical trials and ultimately, commercialization in China. Pineworld is an affiliate of Huapont Life Sciences[2], a $3 Billion dollar pharmaceutical company that specializes in research and development of dermatological and oncological drug products, so they are legitimately-backed and well-equipped to carry out necessary trials.
HSC Trials in Mexico, and the United States
With the finalization of the agreement for R&D and trials in China, Histogen will continue pursuing its goal for an additional $11 million in funding to support a second Phase trial in the US for men and a Phase 1 trial in the US for women. They are hopeful that the data from these trials, and potentially one in Mexico will support an additional Phase 3 trial in the United States by Fall of 2017. Provided the data is positive, an IPO may be in the works by January 2018. These timelines are of course subject to change, but if you are Subscribed[3], you won’t miss any of our updates as the months roll by.
We have confirmed with Histogen that there are no new opportunities on the table regarding partnerships in Japan, and the license from Mexico is still pending.
Histogen Hair Stimulating Complex (HSC)
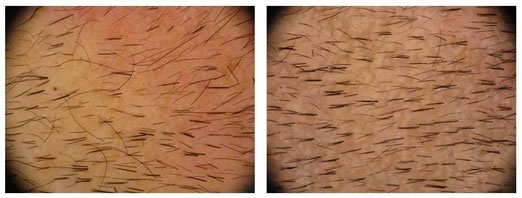
73% Increase in Hair Counts at 1 Year
Hair Stimulating Complex (HSC) is an injectable treatment for hair loss. The injected solution stimulates follicle stem cells and has been shown to encourage significant new hair growth. In trials done thus far, 84.6% of patients receiving just one HSC treatment showed a notable increase in terminal hair count and hair thickness at 12 weeks. The treatment is planned to consist of three injections, so significantly better results are possible in upcoming trial data.
“We are honored to align with Huapont Life Sciences to bring HSC into China,” said Dr. Gail Naughton, Histogen CEO and Chairman of the Board. “With its strong dermatology portfolio, Huapoint is an ideal partner for developing and commercializing Histogen’s unique hair growth product to the world’s most populous nation. Hair loss affects men and women around the world, and our goal is to find the best partners in each region to bring solutions to this global problem.”
HSC has shown excellent safety, and cosmetically significant hair growth in two clinical trials thus far. Their technology process, which mimics the embryonic environment including conditions of low oxygen and suspension, cells triggered to become multipotent naturally produce proteins that are important in the stimulation of hair follicles.
“Huapont Life Sciences is excited to partner with Histogen to bring its innovative product to the millions of men and women in China suffering from hair loss,” said Hayden Zhang, Head of Investments for Huapont and CEO of Pineworld Capital. “By working closely together, we are confident HSC will enjoy tremendous market success.”
Make Sure You’re Subscribed
We encourage you to bookmark our Histogen HSC Project Update[4] page and keep an eye on the list of articles near the bottom for ongoing updates. Better yet, please make sure you are subscribed to our Hair Loss News[5] newsletter, which goes out only once every 2 weeks. You can also customize your preferences[6] and get real-time updates on topics that are of interest to you.
- Histogen – Hair Stimulating Complex Research Update: https://www.hairlosstalk.com/news/new-research/histogen-hsc-interview-0816/
- Huapont Life Sciences: http://api.huapont.cn
- Subscribed: https://www.hairlosstalk.com/subscribe/
- Histogen HSC Project Update: https://www.hairlosstalk.com/pipeline/histogen-hair-stimulating-complex-hsc/
- Hair Loss News: https://www.hairlosstalk.com/subscribe/
- customize your preferences: https://www.hairlosstalk.com/subscribe/
Source URL: https://www.hairlosstalk.com/news/new-research/histogen-hair-stimulating-complex-china/