Treatment of Male Pattern Baldness with Botulinum Toxin: A Pilot Study
Freund, Brian J. D.D.S., M.D.; Schwartz, Marvin D.D.S., M.Sc.
Free Access
Article Outline
 Author Information
Author Information
Crown Institute; Pickering, Ontario, Canada
Correspondence to Dr. Freund, 49 Main Street South, Uxbridge, Ontario L9P 1J4, Canada, freund@crowninstitute.com
This clinical trial has been registered as “Treatment of Male Pattern Baldness with Botulinum Toxin” at http://www.clinicaltrials.gov/ClinicalTrials.gov with identifier NCT00965640.
Back to Top | Article Outline
Sir:

Image Tools
We would like to present the results of an open-label pilot study using botulinum toxin type A (Botox; Allergan, Inc., Irvine, Calif.) for the treatment of androgenetic alopecia. This form of alopecia is believed to be caused by a genetically predisposed sensitivity of hair follicles to the toxic effects of dihydrotestosterone, a metabolite of testosterone. Medical treatment of androgenetic alopecia has previously only been moderately effective using systemic drugs such as finasteride, a 5α-reductase inhibitor.[SUP]1[/SUP]
In this ethically approved study, 50 male subjects aged between 19 and 57 years with Norwood/Hamilton ratings of II to IV participated.[SUP]2[/SUP] The study was 60 weeks in duration, with 12 weeks of run-in followed by two treatment cycles of 24 weeks each. Subjects were injected with 150 units of Botox (5 units per 0.1 ml saline) into the muscles surrounding the scalp, including frontalis, temporalis, periauricular, and occipitalis muscles in equally divided doses over 30 injection sites. The primary outcome measure was a change in hair count in a fixed 2-cm area using a method described by Canfield.[SUP]3[/SUP] Secondary outcome measures included hair loss, measured by having subjects collect loose hair from their pillow with a sticky lint roller, and subjective efficacy using a validated questionnaire. Statistical analysis entailed paired t tests of group means.
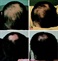
Forty subjects completed the study, and no adverse effects were reported. The treatment response rate was 75 percent. Mean hair counts for the entire group showed a statistically significant (p < 0.0001) increase of 18 percent between baseline and week 48 (Table 1), similar to the results reported with Propecia (Merck, Whitehouse Station, N.J.).[SUP]1[/SUP] Hair regrowth was objectively visible in some subjects (Fig. 1). Secondary outcome measures were also significantly improved. The reduction in hair loss and increase in hair count did not show a statistically significant correlation. This suggested that longer retention of terminal hairs did not account for the increase in hair count.
Mechanistically, the scalp behaves like a drum skin with tensioning muscles around the periphery. These muscle groups—the frontalis, occipitalis, and periauricular muscles and to a minor degree the temporalis—can create a “tight” scalp when chronically active. Because the blood supply to the scalp enters through the periphery, a reduction in blood flow would be most apparent at the distal ends of the vessels, specifically, the vertex and frontal peaks. Areas of the scalp with sparse hair growth have been shown to be relatively hypoxic, have slow capillary refill, and to have high levels of dihydrotestosterone.[SUP]4[/SUP]
Conceptually, Botox “loosens” the scalp, reducing pressure on the perforating vasculature, thereby increasing blood flow and oxygen concentration. The enzymatic conversion of testosterone to dihydrotestosterone is oxygen dependent. In low-oxygen environments, the conversion of testosterone to dihydrotestosterone is favored; whereas in high-oxygen environments, more testosterone is converted to estradiol.[SUP]4[/SUP] Blood flow may therefore be a primary determinant in follicular health. Strategically placed Botox injections appear able to indirectly modify this variable, resulting in reduced hair loss and new hair growth in some men with androgenetic alopecia.
Freund, Brian J. D.D.S., M.D.; Schwartz, Marvin D.D.S., M.Sc.
Free Access
Article Outline
Crown Institute; Pickering, Ontario, Canada
Correspondence to Dr. Freund, 49 Main Street South, Uxbridge, Ontario L9P 1J4, Canada, freund@crowninstitute.com
This clinical trial has been registered as “Treatment of Male Pattern Baldness with Botulinum Toxin” at http://www.clinicaltrials.gov/ClinicalTrials.gov with identifier NCT00965640.
Back to Top | Article Outline
Sir:

Image Tools
We would like to present the results of an open-label pilot study using botulinum toxin type A (Botox; Allergan, Inc., Irvine, Calif.) for the treatment of androgenetic alopecia. This form of alopecia is believed to be caused by a genetically predisposed sensitivity of hair follicles to the toxic effects of dihydrotestosterone, a metabolite of testosterone. Medical treatment of androgenetic alopecia has previously only been moderately effective using systemic drugs such as finasteride, a 5α-reductase inhibitor.[SUP]1[/SUP]
In this ethically approved study, 50 male subjects aged between 19 and 57 years with Norwood/Hamilton ratings of II to IV participated.[SUP]2[/SUP] The study was 60 weeks in duration, with 12 weeks of run-in followed by two treatment cycles of 24 weeks each. Subjects were injected with 150 units of Botox (5 units per 0.1 ml saline) into the muscles surrounding the scalp, including frontalis, temporalis, periauricular, and occipitalis muscles in equally divided doses over 30 injection sites. The primary outcome measure was a change in hair count in a fixed 2-cm area using a method described by Canfield.[SUP]3[/SUP] Secondary outcome measures included hair loss, measured by having subjects collect loose hair from their pillow with a sticky lint roller, and subjective efficacy using a validated questionnaire. Statistical analysis entailed paired t tests of group means.
Forty subjects completed the study, and no adverse effects were reported. The treatment response rate was 75 percent. Mean hair counts for the entire group showed a statistically significant (p < 0.0001) increase of 18 percent between baseline and week 48 (Table 1), similar to the results reported with Propecia (Merck, Whitehouse Station, N.J.).[SUP]1[/SUP] Hair regrowth was objectively visible in some subjects (Fig. 1). Secondary outcome measures were also significantly improved. The reduction in hair loss and increase in hair count did not show a statistically significant correlation. This suggested that longer retention of terminal hairs did not account for the increase in hair count.
Mechanistically, the scalp behaves like a drum skin with tensioning muscles around the periphery. These muscle groups—the frontalis, occipitalis, and periauricular muscles and to a minor degree the temporalis—can create a “tight” scalp when chronically active. Because the blood supply to the scalp enters through the periphery, a reduction in blood flow would be most apparent at the distal ends of the vessels, specifically, the vertex and frontal peaks. Areas of the scalp with sparse hair growth have been shown to be relatively hypoxic, have slow capillary refill, and to have high levels of dihydrotestosterone.[SUP]4[/SUP]
Conceptually, Botox “loosens” the scalp, reducing pressure on the perforating vasculature, thereby increasing blood flow and oxygen concentration. The enzymatic conversion of testosterone to dihydrotestosterone is oxygen dependent. In low-oxygen environments, the conversion of testosterone to dihydrotestosterone is favored; whereas in high-oxygen environments, more testosterone is converted to estradiol.[SUP]4[/SUP] Blood flow may therefore be a primary determinant in follicular health. Strategically placed Botox injections appear able to indirectly modify this variable, resulting in reduced hair loss and new hair growth in some men with androgenetic alopecia.